10-Q
falseQ2--12-310001539029http://fasb.org/us-gaap/2025#LicenseMemberhttp://fasb.org/us-gaap/2025#LicenseMemberhttp://fasb.org/us-gaap/2025#LicenseMemberhttp://fasb.org/us-gaap/2025#LicenseMember0001539029us-gaap:AdditionalPaidInCapitalMember2024-04-012024-06-300001539029clsd:XipereMemberclsd:BauschHealthIrelandLimitedMember2025-01-012025-06-300001539029us-gaap:RestrictedStockUnitsRSUMember2024-01-012024-06-300001539029us-gaap:RetainedEarningsMember2025-06-300001539029us-gaap:MachineryAndEquipmentMember2025-06-300001539029us-gaap:RestrictedStockUnitsRSUMember2024-04-012024-06-300001539029clsd:BiocrystPharmaceuticalsIncMemberclsd:LicenseArrangementMembersrt:MaximumMember2023-11-010001539029clsd:XipereMemberclsd:BauschHealthIrelandLimitedMembersrt:MaximumMember2019-10-222019-10-2200015390292025-04-012025-06-300001539029clsd:BiocrystPharmaceuticalsIncMemberclsd:LicenseArrangementMembersrt:MaximumMember2023-11-012023-11-010001539029us-gaap:CommonStockMemberclsd:InMayTwoThousandTwentyThreeMemberclsd:CantorFitzgeraldCo1Member2025-06-300001539029us-gaap:RetainedEarningsMember2023-12-310001539029us-gaap:AdditionalPaidInCapitalMember2024-12-310001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-04-012024-06-3000015390292025-03-310001539029clsd:LicenseArrangementMemberclsd:ArcticVisionsLimitedMember2021-09-012021-09-300001539029us-gaap:CommonStockMemberclsd:InMayTwoThousandTwentyThreeMemberclsd:CantorFitzgeraldCo1Member2025-01-012025-06-300001539029us-gaap:RetainedEarningsMember2024-03-3100015390292025-01-012025-03-310001539029us-gaap:RetainedEarningsMember2024-04-012024-06-300001539029us-gaap:RetainedEarningsMember2024-06-300001539029us-gaap:MeasurementInputExpectedDividendRateMemberus-gaap:WarrantMember2025-06-300001539029clsd:CommonStockWarrantsMember2024-02-060001539029us-gaap:GeneralAndAdministrativeExpenseMemberclsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2024-04-012024-06-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2025-04-012025-06-300001539029us-gaap:RetainedEarningsMember2024-01-012024-03-310001539029us-gaap:WarrantMemberus-gaap:MeasurementInputExpectedTermMember2025-06-300001539029us-gaap:RetainedEarningsMember2024-12-310001539029stpr:GA2024-10-012024-10-010001539029clsd:RoyaltySubMemberMemberclsd:RoyaltyPurchaseAndSaleAgreementMembersrt:MaximumMember2022-08-082022-08-080001539029us-gaap:RelatedPartyMemberclsd:AuraBiosciencesMember2025-04-012025-06-300001539029us-gaap:CommonStockMember2024-12-3100015390292024-01-012024-12-310001539029us-gaap:RetainedEarningsMember2025-04-012025-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2025-01-012025-06-300001539029us-gaap:WarrantMemberus-gaap:MeasurementInputExercisePriceMember2025-06-300001539029us-gaap:ComputerEquipmentMember2025-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2025-04-012025-06-300001539029us-gaap:AdditionalPaidInCapitalMember2025-06-300001539029stpr:GA2022-11-012022-11-300001539029clsd:NonVestedRestrictedStockUnitsMember2024-01-012024-06-300001539029us-gaap:FurnitureAndFixturesMember2025-06-300001539029us-gaap:CommonStockMember2025-01-012025-03-3100015390292024-06-300001539029us-gaap:CommonStockMember2024-02-062024-02-060001539029srt:MaximumMember2016-09-300001539029clsd:CommonStockWarrantsMember2016-09-012016-09-300001539029us-gaap:RestrictedStockUnitsRSUMember2025-06-300001539029us-gaap:WarrantMemberus-gaap:MeasurementInputConversionPriceMember2025-06-3000015390292025-06-300001539029us-gaap:EmployeeStockOptionMember2024-01-012024-06-300001539029us-gaap:WarrantMemberus-gaap:MeasurementInputRiskFreeInterestRateMember2025-06-300001539029srt:MinimumMemberclsd:ArcticVisionsLimitedMember2020-03-102020-03-100001539029us-gaap:EmployeeStockOptionMember2025-06-300001539029us-gaap:RetainedEarningsMember2025-01-012025-03-3100015390292024-02-102024-12-310001539029us-gaap:CommonStockMember2025-04-012025-06-300001539029srt:ScenarioForecastMember2025-07-012025-09-300001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:GeneralAndAdministrativeExpenseMember2025-01-012025-06-300001539029clsd:BiocrystPharmaceuticalsIncMemberclsd:LicenseArrangementMember2023-11-010001539029us-gaap:CommonStockMemberclsd:InMayTwoThousandTwentyThreeMemberclsd:CantorFitzgeraldCo1Member2024-06-300001539029us-gaap:RelatedPartyMemberclsd:BiocrystLicenseAgreementMember2024-04-012024-06-300001539029us-gaap:RelatedPartyMemberus-gaap:AccruedLiabilitiesMember2024-12-310001539029us-gaap:CommonStockMember2023-12-310001539029clsd:RoyaltyPurchaseAndSaleAgreementMember2022-08-082022-08-080001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-04-012025-06-300001539029stpr:GA2022-11-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-06-300001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:GeneralAndAdministrativeExpenseMember2024-01-012024-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-04-012025-06-300001539029us-gaap:CommonStockMember2024-01-012024-03-310001539029us-gaap:RelatedPartyMember2024-04-012024-06-300001539029us-gaap:CommonStockMember2025-03-310001539029clsd:NonVestedRestrictedStockUnitsMember2025-01-012025-06-300001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-01-012024-06-300001539029us-gaap:RelatedPartyMemberclsd:BiocrystLicenseAgreementMember2025-01-012025-06-300001539029us-gaap:GeneralAndAdministrativeExpenseMemberus-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2025-01-012025-06-300001539029clsd:UpfrontPaymentMemberclsd:BiocrystPharmaceuticalsIncMemberclsd:LicenseArrangementMember2023-11-012023-11-010001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:GeneralAndAdministrativeExpenseMember2025-04-012025-06-300001539029us-gaap:LeaseholdImprovementsMember2024-12-310001539029us-gaap:RelatedPartyMemberclsd:AuraBiosciencesMember2025-01-012025-06-300001539029us-gaap:WarrantMember2024-02-060001539029clsd:RoyaltySubMemberMemberclsd:RoyaltyPurchaseAndSaleAgreementMember2022-08-082022-08-080001539029clsd:LicenseArrangementMembersrt:MaximumMemberclsd:ArcticVisionsLimitedMember2020-03-102020-03-100001539029us-gaap:ComputerEquipmentMember2024-12-310001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2025-06-3000015390292024-02-062024-02-060001539029us-gaap:AdditionalPaidInCapitalMember2025-04-012025-06-300001539029srt:MaximumMember2025-05-3000015390292022-08-082022-08-080001539029us-gaap:RelatedPartyMemberclsd:BiocrystLicenseAgreementMember2025-04-012025-06-300001539029us-gaap:GeneralAndAdministrativeExpenseMemberclsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2025-04-012025-06-300001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-04-012025-06-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2024-04-012024-06-300001539029us-gaap:CommonStockMemberclsd:InMayTwoThousandTwentyThreeMemberclsd:CantorFitzgeraldCo1Member2024-01-012024-06-300001539029us-gaap:AdditionalPaidInCapitalMember2025-03-310001539029us-gaap:MachineryAndEquipmentMember2024-12-3100015390292024-01-012024-06-300001539029us-gaap:RestrictedStockUnitsRSUMember2025-01-012025-06-3000015390292025-01-012025-06-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-01-012024-06-300001539029clsd:AccumulatedOtherComprehensiveLossIncomeMember2024-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2024-01-012024-06-300001539029us-gaap:RestrictedStockUnitsRSUMember2025-04-012025-06-300001539029clsd:AtTheMarketSalesAgreementMemberclsd:InMayTwoThousandTwentyThreeMemberclsd:CantorFitzgeraldCo1Member2023-05-310001539029clsd:AccumulatedOtherComprehensiveLossIncomeMember2024-04-012024-06-300001539029us-gaap:WarrantMember2024-01-012024-06-300001539029us-gaap:EmployeeStockOptionMember2025-01-012025-06-300001539029clsd:UpfrontPaymentMemberclsd:LicenseArrangementMemberclsd:ArcticVisionsLimitedMember2020-03-102020-03-100001539029us-gaap:AdditionalPaidInCapitalMember2023-12-3100015390292024-12-310001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2025-01-012025-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-01-012024-06-300001539029us-gaap:RelatedPartyMemberus-gaap:AccountsPayableMember2024-12-310001539029us-gaap:AdditionalPaidInCapitalMember2024-01-012024-03-310001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2024-01-012024-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2024-04-012024-06-3000015390292024-01-012024-03-310001539029us-gaap:AdditionalPaidInCapitalMember2024-06-300001539029us-gaap:CommonStockMember2025-06-300001539029us-gaap:WarrantMemberus-gaap:MeasurementInputPriceVolatilityMember2025-06-300001539029us-gaap:RetainedEarningsMember2025-03-310001539029us-gaap:RelatedPartyMemberclsd:BiocrystLicenseAgreementMember2024-01-012024-06-300001539029us-gaap:WarrantMembersrt:MaximumMember2024-02-060001539029us-gaap:AdditionalPaidInCapitalMember2024-03-310001539029clsd:WorkInProcessMember2024-12-310001539029us-gaap:AdditionalPaidInCapitalMember2025-01-012025-03-310001539029us-gaap:GeneralAndAdministrativeExpenseMemberus-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2024-01-012024-06-300001539029clsd:CommonStockWarrantsMember2016-09-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2025-01-012025-06-300001539029clsd:WorkInProcessMember2025-06-300001539029us-gaap:CommonStockMember2024-03-310001539029us-gaap:GeneralAndAdministrativeExpenseMemberus-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2024-04-012024-06-3000015390292023-12-310001539029us-gaap:WarrantMember2025-01-012025-06-300001539029us-gaap:GeneralAndAdministrativeExpenseMemberus-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2025-04-012025-06-300001539029us-gaap:CommonStockMembersrt:MaximumMember2024-02-062024-02-0600015390292025-08-080001539029us-gaap:EmployeeStockOptionMember2025-01-012025-06-300001539029clsd:BauschHealthIrelandLimitedMember2019-10-222019-10-220001539029srt:MinimumMember2025-05-300001539029stpr:GA2024-02-012024-02-2900015390292024-04-012024-06-300001539029us-gaap:GeneralAndAdministrativeExpenseMemberclsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2024-01-012024-06-3000015390292024-03-310001539029srt:MaximumMemberclsd:ArcticVisionsLimitedMember2020-03-102020-03-1000015390292025-07-012025-09-300001539029us-gaap:FurnitureAndFixturesMember2024-12-3100015390292024-02-092024-02-090001539029us-gaap:RestrictedStockUnitsRSUMember2024-12-310001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMember2024-12-310001539029us-gaap:LeaseholdImprovementsMember2025-06-300001539029us-gaap:RelatedPartyMember2024-01-012024-06-300001539029us-gaap:EmployeeStockOptionMemberclsd:TwoThousandsAndElevenStockIncentivePlanTwoThousandsAndSixteenEquityIncentivePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-04-012024-06-300001539029us-gaap:RestrictedStockUnitsRSUMemberus-gaap:GeneralAndAdministrativeExpenseMember2024-04-012024-06-300001539029us-gaap:LeaseholdImprovementsMember2025-01-012025-06-300001539029us-gaap:WarrantMember2024-02-062024-02-060001539029us-gaap:GeneralAndAdministrativeExpenseMemberclsd:TwoThousandSixteenEmployeeStockPurchasePlanMember2025-01-012025-06-300001539029us-gaap:CommonStockMember2024-06-300001539029clsd:TwoThousandSixteenEmployeeStockPurchasePlanMemberus-gaap:ResearchAndDevelopmentExpenseMember2024-04-012024-06-30xbrli:pureutr:sqftxbrli:sharesiso4217:USDxbrli:sharesiso4217:USD
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-Q
(Mark One)
☒ QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the quarterly period ended June 30, 2025
OR
☐ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
Commission File Number: 001-37783
Clearside Biomedical, Inc.
(Exact Name of Registrant as Specified in its Charter)
|
|
Delaware |
45-2437375 |
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer
Identification No.) |
900 North Point Parkway, Suite 200 Alpharetta, GA |
30005 |
(Address of principal executive offices) |
(Zip Code) |
(678) 270-3631
Registrant’s telephone number, including area code
N/A
(Former name, former address and former fiscal year, if changed since last report)
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
Common Stock, par value $0.001 per share |
CLSD |
The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
|
|
|
|
|
|
Large accelerated filer |
|
☐ |
|
Accelerated filer |
|
☐ |
|
|
|
|
Non-accelerated filer |
|
☒ |
|
Smaller reporting company |
|
☒ |
|
|
|
|
|
|
|
|
|
|
|
Emerging growth company |
|
☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
As of August 8, 2025, the registrant had 78,510,318 shares of common stock, $0.001 par value per share, outstanding.
PART I – FINANCIAL INFORMATION
Item 1. Financial Statements
CLEARSIDE BIOMEDICAL, INC.
Consolidated Balance Sheets
(in thousands, except share and per share data)
(unaudited)
|
|
|
|
|
|
|
|
|
|
|
June 30,
2025 |
|
|
December 31,
2024 |
|
Assets |
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
9,376 |
|
|
$ |
20,020 |
|
Accounts receivable |
|
|
816 |
|
|
|
507 |
|
Prepaid expenses |
|
|
1,580 |
|
|
|
734 |
|
Other current assets |
|
|
13 |
|
|
|
13 |
|
Total current assets |
|
|
11,785 |
|
|
|
21,274 |
|
Property and equipment, net |
|
|
3,065 |
|
|
|
3,225 |
|
Operating lease right-of-use asset |
|
|
452 |
|
|
|
597 |
|
Other assets |
|
|
30 |
|
|
|
30 |
|
Total assets |
|
$ |
15,332 |
|
|
$ |
25,126 |
|
Liabilities and stockholders’ deficit |
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
Accounts payable (includes $491 to a related party as of
December 31, 2024) |
|
$ |
1,129 |
|
|
$ |
1,452 |
|
Accrued liabilities (includes $304 to a related party as of
December 31, 2024) |
|
|
1,537 |
|
|
|
2,967 |
|
Current portion of operating lease liabilities |
|
|
381 |
|
|
|
375 |
|
Total current liabilities |
|
|
3,047 |
|
|
|
4,794 |
|
Liability related to the sales of future royalties, net |
|
|
55,702 |
|
|
|
51,767 |
|
Warrant liabilities |
|
|
4,765 |
|
|
|
6,692 |
|
Operating lease liabilities |
|
|
152 |
|
|
|
328 |
|
Other non-current liabilities |
|
|
400 |
|
|
|
400 |
|
Total liabilities |
|
|
64,066 |
|
|
|
63,981 |
|
Commitments and contingencies |
|
|
|
|
|
|
Stockholders’ deficit: |
|
|
|
|
|
|
Preferred stock, $0.001 par value; 10,000,000 shares authorized and no
shares issued at June 30, 2025 and December 31, 2024 |
|
|
— |
|
|
|
— |
|
Common stock, $0.001 par value; 400,000,000 and 200,000,000 shares authorized
at June 30, 2025 and December 31, 2024, respectively; 78,495,760 and 76,578,383
shares issued and outstanding at June 30, 2025 and December 31, 2024, respectively |
|
|
78 |
|
|
|
77 |
|
Additional paid-in capital |
|
|
319,181 |
|
|
|
316,343 |
|
Accumulated deficit |
|
|
(367,993 |
) |
|
|
(355,275 |
) |
Total stockholders’ deficit |
|
|
(48,734 |
) |
|
|
(38,855 |
) |
Total liabilities and stockholders’ deficit |
|
$ |
15,332 |
|
|
$ |
25,126 |
|
See accompanying notes to the consolidated financial statements.
CLEARSIDE BIOMEDICAL, INC.
Consolidated Statements of Operations
(in thousands, except share and per share data)
(unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
License and other revenue (includes $442 and $81
from a related party for the three months ended
June 30, 2025 and 2024, respectively, and $448
and $156 for the six months ended June 30,
2025 and 2024, respectively) |
|
$ |
492 |
|
|
$ |
90 |
|
|
$ |
2,822 |
|
|
$ |
320 |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
Cost of goods sold |
|
|
— |
|
|
|
— |
|
|
|
248 |
|
|
|
— |
|
Research and development (includes $243 and $503 to a
related party for the three and six months ended
June 30, 2024, respectively) |
|
|
1,584 |
|
|
|
4,603 |
|
|
|
6,047 |
|
|
|
10,218 |
|
General and administrative |
|
|
2,476 |
|
|
|
3,077 |
|
|
|
5,300 |
|
|
|
5,901 |
|
Total operating expenses |
|
|
4,060 |
|
|
|
7,680 |
|
|
|
11,595 |
|
|
|
16,119 |
|
Loss from operations |
|
|
(3,568 |
) |
|
|
(7,590 |
) |
|
|
(8,773 |
) |
|
|
(15,799 |
) |
Interest income |
|
|
115 |
|
|
|
419 |
|
|
|
278 |
|
|
|
767 |
|
Other income, net |
|
|
1,720 |
|
|
|
1,917 |
|
|
|
1,927 |
|
|
|
418 |
|
Non-cash interest expense on liability
related to the sales of future royalties |
|
|
(2,762 |
) |
|
|
(2,340 |
) |
|
|
(5,435 |
) |
|
|
(4,743 |
) |
Loss before income taxes |
|
|
(4,495 |
) |
|
|
(7,594 |
) |
|
|
(12,003 |
) |
|
|
(19,357 |
) |
Income tax expense |
|
|
— |
|
|
|
— |
|
|
|
715 |
|
|
|
— |
|
Net loss |
|
$ |
(4,495 |
) |
|
$ |
(7,594 |
) |
|
$ |
(12,718 |
) |
|
$ |
(19,357 |
) |
Net loss per share of common stock — basic and diluted |
|
$ |
(0.06 |
) |
|
$ |
(0.10 |
) |
|
$ |
(0.16 |
) |
|
$ |
(0.27 |
) |
Weighted average shares outstanding — basic and diluted |
|
|
77,844,055 |
|
|
|
74,731,139 |
|
|
|
77,385,497 |
|
|
|
72,292,183 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
$ |
(4,495 |
) |
|
$ |
(7,594 |
) |
|
$ |
(12,718 |
) |
|
$ |
(19,357 |
) |
Unrealized loss on available-for-sale investments |
|
|
— |
|
|
|
(1 |
) |
|
|
— |
|
|
|
(1 |
) |
Comprehensive loss |
|
$ |
(4,495 |
) |
|
$ |
(7,595 |
) |
|
$ |
(12,718 |
) |
|
$ |
(19,358 |
) |
See accompanying notes to the consolidated financial statements.
CLEARSIDE BIOMEDICAL, INC.
Consolidated Statements of Stockholders’ Deficit
(in thousands, except share data)
(unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended June 30, 2025 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
|
|
|
Common Stock |
|
|
Additional |
|
|
Accumulated |
|
|
Stockholders' |
|
|
|
Shares |
|
|
Amount |
|
|
Paid-In-Capital |
|
|
Deficit |
|
|
Deficit |
|
Balance at December 31, 2024 |
|
|
76,578,383 |
|
|
$ |
77 |
|
|
$ |
316,343 |
|
|
$ |
(355,275 |
) |
|
$ |
(38,855 |
) |
Issuance of common stock under at-the-market
sales agreement |
|
|
426,822 |
|
|
|
— |
|
|
|
434 |
|
|
|
— |
|
|
|
434 |
|
Vesting and settlement of restricted stock units |
|
|
251,359 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Issuance of common stock under employee stock
purchase plan |
|
|
16,222 |
|
|
|
— |
|
|
|
13 |
|
|
|
— |
|
|
|
13 |
|
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
721 |
|
|
|
— |
|
|
|
721 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(8,223 |
) |
|
|
(8,223 |
) |
Balance at March 31, 2025 |
|
|
77,272,786 |
|
|
|
77 |
|
|
|
317,511 |
|
|
|
(363,498 |
) |
|
|
(45,910 |
) |
Issuance of common stock under at-the-market
sales agreement |
|
|
1,222,974 |
|
|
|
1 |
|
|
|
976 |
|
|
|
— |
|
|
|
977 |
|
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
694 |
|
|
|
— |
|
|
|
694 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(4,495 |
) |
|
|
(4,495 |
) |
Balance at June 30, 2025 |
|
|
78,495,760 |
|
|
$ |
78 |
|
|
$ |
319,181 |
|
|
$ |
(367,993 |
) |
|
$ |
(48,734 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended June 30, 2024 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other |
|
|
Total |
|
|
|
Common Stock |
|
|
Additional |
|
|
Accumulated |
|
|
Comprehensive |
|
|
Stockholders' |
|
|
|
Shares |
|
|
Amount |
|
|
Paid-In-Capital |
|
|
Deficit |
|
|
Loss |
|
|
Deficit |
|
Balance at December 31, 2023 |
|
|
62,850,841 |
|
|
$ |
63 |
|
|
$ |
304,948 |
|
|
$ |
(320,923 |
) |
|
$ |
— |
|
|
$ |
(15,912 |
) |
Issuance of common stock under registered direct
offering |
|
|
11,111,111 |
|
|
|
11 |
|
|
|
4,309 |
|
|
|
— |
|
|
|
— |
|
|
|
4,320 |
|
Issuance of common stock under at-the-market
sales agreement |
|
|
339,912 |
|
|
|
— |
|
|
|
450 |
|
|
|
— |
|
|
|
— |
|
|
|
450 |
|
Exercise of stock options |
|
|
10,000 |
|
|
|
— |
|
|
|
12 |
|
|
|
— |
|
|
|
— |
|
|
|
12 |
|
Vesting and settlement of restricted stock units |
|
|
397,594 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Issuance of common stock under employee
stock purchase plan |
|
|
21,681 |
|
|
|
— |
|
|
|
21 |
|
|
|
— |
|
|
|
— |
|
|
|
21 |
|
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
1,062 |
|
|
|
— |
|
|
|
— |
|
|
|
1,062 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(11,763 |
) |
|
|
— |
|
|
|
(11,763 |
) |
Balance at March 31, 2024 |
|
|
74,731,139 |
|
|
|
74 |
|
|
|
310,802 |
|
|
|
(332,686 |
) |
|
|
— |
|
|
|
(21,810 |
) |
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
1,120 |
|
|
|
— |
|
|
|
— |
|
|
|
1,120 |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(7,594 |
) |
|
|
— |
|
|
|
(7,594 |
) |
Other comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(1 |
) |
|
|
(1 |
) |
Balance at June 30, 2024 |
|
|
74,731,139 |
|
|
$ |
74 |
|
|
$ |
311,922 |
|
|
$ |
(340,280 |
) |
|
$ |
(1 |
) |
|
$ |
(28,285 |
) |
See accompanying notes to the consolidated financial statements.
CLEARSIDE BIOMEDICAL, INC.
Consolidated Statements of Cash Flows
(in thousands)
(unaudited)
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
Operating activities |
|
|
|
|
|
|
Net loss |
|
$ |
(12,718 |
) |
|
$ |
(19,357 |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
Non-cash interest expense on liability related to the sales of
future royalties, net of issuance costs accretion |
|
|
5,435 |
|
|
|
4,743 |
|
Depreciation |
|
|
169 |
|
|
|
72 |
|
Share-based compensation expense |
|
|
1,415 |
|
|
|
2,182 |
|
Change in fair value of warrant liabilities |
|
|
(1,927 |
) |
|
|
(1,206 |
) |
Issuance costs allocated to warrant liabilities |
|
|
— |
|
|
|
787 |
|
Amortization and accretion of available-for-sales investments, net |
|
|
— |
|
|
|
(109 |
) |
Changes in operating assets and liabilities: |
|
|
|
|
|
|
Prepaid expenses and other current assets |
|
|
(1,155 |
) |
|
|
795 |
|
Other assets and liabilities |
|
|
(25 |
) |
|
|
(497 |
) |
Accounts payable and accrued liabilities (includes $254 to a related party
for the six months ended June 30, 2024) |
|
|
(1,753 |
) |
|
|
(1,091 |
) |
Deferred revenue |
|
|
— |
|
|
|
150 |
|
Net cash used in operating activities |
|
|
(10,559 |
) |
|
|
(13,531 |
) |
Investing activities |
|
|
|
|
|
|
Acquisition of property and equipment |
|
|
(9 |
) |
|
|
(480 |
) |
Purchase of short-term investments |
|
|
— |
|
|
|
(11,014 |
) |
Net cash used in investing activities |
|
|
(9 |
) |
|
|
(11,494 |
) |
Financing activities |
|
|
|
|
|
|
Proceeds from issuance of common stock and warrants under
registered direct offering, net of issuance costs |
|
|
— |
|
|
|
13,860 |
|
Proceeds from at-the-market sales agreement, net of issuance costs |
|
|
1,411 |
|
|
|
450 |
|
Payments on royalty purchase and sale agreement |
|
|
(1,500 |
) |
|
|
— |
|
Proceeds from exercise of stock options |
|
|
— |
|
|
|
12 |
|
Proceeds from shares issued under employee stock purchase plan |
|
|
13 |
|
|
|
21 |
|
Net cash (used in) provided by financing activities |
|
|
(76 |
) |
|
|
14,343 |
|
Net decrease in cash and cash equivalents |
|
|
(10,644 |
) |
|
|
(10,682 |
) |
Cash and cash equivalents, beginning of period |
|
|
20,020 |
|
|
|
28,920 |
|
Cash and cash equivalent, end of period |
|
$ |
9,376 |
|
|
$ |
18,238 |
|
Supplemental disclosure |
|
|
|
|
|
|
Purchase of property and equipment included in accrued liabilities |
|
$ |
— |
|
|
$ |
4 |
|
See accompanying notes to the consolidated financial statements.
CLEARSIDE BIOMEDICAL, INC.
Notes to the Consolidated Financial Statements
(unaudited)
1. The Company
Clearside Biomedical, Inc. (the “Company”) is a biopharmaceutical company focused on revolutionizing the delivery of therapies to the back of the eye through the suprachoroidal space (SCS®). Incorporated in the State of Delaware on May 26, 2011, the Company has its corporate headquarters in Alpharetta, Georgia.
The Company’s activities since inception have primarily consisted of developing product and technology rights, raising capital and performing research and development activities. The Company is subject to a number of risks and uncertainties similar to those of other life science companies at a similar stage of development, including, among others, the need to obtain adequate additional financing, successful development efforts including regulatory approval of products, compliance with government regulations, successful commercialization of potential products, protection of proprietary technology and dependence on key individuals.
Liquidity
The Company had cash and cash equivalents of $9.4 million as of June 30, 2025. In July 2025, the Company announced plans to explore a full range of strategic alternatives to advance its SCS platform and drug development pipeline to maximize stockholder value. The Company has retained Piper Sandler, a leading investment bank with substantial experience in the biotechnology industry, to support it with the strategic evaluation process. Strategic alternatives under consideration include the sale, license, monetization and/or divestiture of one or more of the Company’s assets and technologies, collaboration, partnership, merger, acquisition, joint ventures, or other strategic transactions. If a strategic alternative is not available, the Company will be required to take additional actions to fund the Company’s operations, or it may be forced to file for bankruptcy or wind down its operations. In addition, in July 2025, the Company implemented a plan pursuant to which all of the Company’s employees had their employment with the Company terminated and transitioned into consulting roles with the Company (the "Reduction in Force"). The Company estimates that it will incur charges of approximately $2.6 million for severance and other employee termination related costs in the third quarter of 2025. As part of the strategic alternative process, the Company has paused all internal research and development programs which can be resumed if the Company is able to raise additional capital.
Historically, the Company has funded its operations primarily through the sale of common stock and convertible preferred stock, the issuance of warrants, the issuance of long-term debt, and license agreements.
On February 6, 2024, the Company entered into a securities purchase agreement with institutional investors and an existing stockholder, pursuant to which the Company issued and sold, in a registered direct offering (the “Registered Direct Offering”): (i) an aggregate of 11,111,111 shares of its common stock; and (ii) warrants to purchase up to 11,111,111 shares of common stock (the “Warrants”). The combined purchase price of each share and accompanying Warrant was $1.35. The exercise price for the Warrants is $1.62 per share. The Warrants are currently exercisable and will expire on August 9, 2029. The net proceeds to the Company from the Registered Direct Offering were $13.9 million.
On January 31, 2024 (the “Amendment Effective Date”), the Company entered into a fourth amendment to the license agreement (as amended, the “Emory License Agreement”) with Emory University and Georgia Tech Research Corporation (collectively, the “Licensor”) pursuant to which the parties agreed to reduce the Sublicense Percentage (as defined in the Emory License Agreement) from a low double digit percentage to a high single digit percentage that the Company will pay the Licensor applicable to any fees or payments paid to the Company by any Sublicensee (as defined in the Emory License Agreement) of the Licensed Patents and/or Licensed Technology (each as defined in the Emory License Agreement), excluding (i) amounts paid to the Company by a Sublicensee to reimburse the Company for certain research and development costs pursuant to a written agreement between the Company and such Sublicensee, (ii) the value of intellectual property transferred or granted to the Company if necessary or helpful to the development or commercialization of Licensed Products (as defined in the Emory License Agreement) and (iii) amounts paid for shares of the Company’s stock. The payment to Licensor of any such Sublicense Percentage is due within 30 days of receipt by the Company of a qualifying payment from a Sublicensee, provided however, with respect to any qualifying payments received by the Company from a Sublicensee after July 1, 2023 but prior to January 1, 2025, the payment to Licensor of any such Sublicensee Percentage was due to Licensor by March 31, 2025. The parties also agreed to a revised annual license maintenance fee due each year (the “Maintenance Fee”) starting in 2023 through 2028, as follows: $0.3 million for 2023 through 2025, $0.4 million for 2026 and for 2027 and $0.5 million for 2028. The Company paid the Maintenance Fee for 2023 in February 2024 and the Maintenance Fee for 2024 in October 2024. The remaining annual Maintenance Fee payments are due on October 1st of each year.
In May 2023, the Company entered into a Controlled Equity OfferingSM Sales Agreement (the "Sales Agreement") with Cantor Fitzgerald & Co. ("Cantor") under which the Company may offer and sell, from time to time at its sole discretion, shares of its common stock, having an aggregate offering price of up to $50.0 million through Cantor as its sales agent. During the six months ended June 30, 2025, the Company sold 1,649,796 shares of its common stock for net proceeds of $1.4 million under the Sales
Agreement. During the six months ended June 30, 2024, the Company sold 339,912 shares of its common stock under the Sales Agreement for net proceeds of $0.5 million.
The Company has suffered recurring losses and negative cash flows from operations since inception and anticipates incurring additional losses until such time, if ever, that it can generate significant revenue. The Company has no current source of revenue to sustain present activities. The Company does not expect to generate other meaningful revenue until and unless the Company's licensees successfully commercialize XIPERE and the Company has fulfilled its obligations under the Purchase and Sale Agreement, its other licensees receive regulatory approval and successfully commercialize its product candidates, or the Company commercializes its product candidates either on its own or with a third party. In the absence of product or other revenues, the amount, timing, nature or source of which cannot be predicted, the Company’s losses will continue as it conducts its research and development activities.
The Company will continue to need to obtain additional financing to fund future operations, including completing the development, partnering and potential commercialization of its primary product candidates. The Company will need to obtain financing to complete the development and conduct clinical trials for the regulatory approval of its product candidates if requested by regulatory bodies. If such product candidates were to receive regulatory approval, the Company would need to obtain financing to prepare for the potential commercialization of its product candidates, if the Company decides to commercialize the products on its own.
These conditions raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date the financial statements are issued. Based on its current plans and forecasted expenses, the Company does not expect its cash and cash equivalents will enable the Company to fund its planned operating expenses and capital expenditure requirements for the next 12 months from the date of this filing.
Accordingly, the Company’s consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might result should the Company be unable to continue as a going concern.
Until the Company can generate sufficient revenue, the Company will need to finance future cash needs through public or private equity offerings, license agreements, debt financings or restructurings, collaborations, strategic alliances and marketing or distribution arrangements.
2. Significant Accounting Policies
Basis of Presentation and Principles of Consolidation
The Company's consolidated financial statements include the results of the financial operations of Clearside Biomedical, Inc. and Royalty Sub, which was formed for the purposes of the transactions contemplated by the Purchase and Sale Agreement described in Note 5. All intercompany balances and transactions have been eliminated.
The Company’s consolidated financial statements have been prepared in conformity with U.S. generally accepted accounting principles (“U.S. GAAP”). In the opinion of management, the Company has made all necessary adjustments, which include normal recurring adjustments necessary for a fair statement of the Company’s consolidated financial position and results of operations for the interim periods presented. The results for the three and six months ended June 30, 2025 are not indicative of results to be expected for the full year ending December 31, 2025, any other interim periods or any future year or period. These unaudited consolidated financial statements should be read in conjunction with the audited consolidated financial statements and related footnotes, which are included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2024, filed with the Securities and Exchange Commission on March 27, 2025.
During the six months ended June 30, 2025, the Company recorded an immaterial out of period adjustment of $641,000 in income tax expense and accrued liabilities related to a foreign jurisdiction.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and reported amounts of income and expenses during the reporting periods. Significant items subject to such estimates and assumptions include the estimate of the total amount of future royalty revenue and milestone payments to be generated over the life of the Purchase and Sale Agreement described in Note 5, certain assumptions used in the valuation of warrant liabilities, revenue recognition, the accounting for useful lives to calculate depreciation and amortization, clinical trial expense accruals, share-based compensation expense and income tax valuation allowance. Actual results could differ from these estimates.
Revenue Recognition
The Company recognizes revenue from its contracts with customers under Financial Accounting Standards Board ("FASB") Accounting Standards Codification (“ASC”) 606, Revenue from Contracts with Customers. The Company’s primary revenue arrangements are license agreements, which typically include upfront payments, regulatory and commercial milestone payments and royalties based on future product sales. The arrangements may also include payments for the Company’s SCS Microinjector devices as well as payments for assistance and oversight of the customer’s use of the Company’s technology. In determining the amount of revenue to be recognized under these agreements, the Company performs the following steps: (i) identifies the promised goods and services to be transferred in the contract, (ii) identifies the performance obligations, (iii) determines the transaction price, (iv) allocates the transaction price to the performance obligations and (v) recognizes revenue as the performance obligations are satisfied.
The Company receives payments from its customers based on billing schedules established in each contract. Upfront and other payments may require deferral of revenue recognition to a future period until the Company performs its obligations under the arrangement. Amounts are recorded as accounts receivable when the Company’s right to consideration is unconditional. The Company does not assess whether a contract has a significant financing component if the expectation at contract inception is such that the period between payment by the customer and the transfer of the promised goods or services to the customer will be one year or less.
Research and Development Costs
Research and development costs are charged to expense as incurred and include:
•employee-related expenses, including salaries, benefits, travel and share-based compensation expense for research and development personnel;
•expenses incurred under agreements with contract research organizations, contract manufacturing organizations and consultants that conduct preclinical studies and clinical trials;
•costs associated with preclinical and clinical development activities;
•costs associated with submitting regulatory approval applications for the Company’s product candidates;
•costs associated with training physicians on the suprachoroidal injection procedure and educating and providing them with appropriate product candidate information;
•costs associated with technology and intellectual property licenses;
•costs for the Company’s research and development facility; and
•depreciation expense for assets used in research and development activities.
Costs for certain development activities, such as clinical trial activities, are recognized based on an evaluation of the estimated total costs for the clinical trial, progress to completion of specific tasks using data such as participant enrollment, pass-through expenses, clinical site activations, data from the clinical sites or information provided to the Company by its vendors on their actual costs incurred. Payments for these activities are based on the terms of the individual contracts and any subsequent amendments, which may differ from the patterns of costs incurred, and are reflected in the consolidated financial statements as prepaid expenses or accrued liabilities.
Share-Based Compensation
Compensation cost related to share-based awards granted to employees, directors and consultants is measured based on the estimated fair value of the award at the grant date. The Company estimates the fair value of stock options using a Black-Scholes option pricing model. The fair value of restricted stock units granted is measured based on the market value of the Company’s common stock on the date of grant. Share-based compensation costs are expensed on a straight-line basis over the relevant vesting period. The Reduction in Force implemented in July 2025 did not change the vesting terms and conditions of the share-based awards.
Compensation cost related to shares purchased through the Company’s employee stock purchase plan, which is considered compensatory, is based on the estimated fair value of the shares on the offering date, including consideration of the discount and the look-back period. The Company estimates the fair value of the shares using a Black-Scholes option pricing model. Compensation expense is recognized over the six-month withholding period prior to the purchase date.
All share-based compensation costs are recorded in general and administrative or research and development costs in the consolidated statements of operations based upon the recipient's underlying role within the Company.
Cash Equivalents
Cash equivalents consist of short-term, highly liquid investments with an original term of three months or less at the date of purchase.
Concentration of Credit Risk Arising From Cash Deposits in Excess of Insured Limits
The Company maintains its cash in bank deposits that at times may exceed federally insured limits. The Company has not experienced any loss in such accounts. The Company believes it is not exposed to any significant risks with respect to its cash balances.
Liability Related to the Sales of Future Royalties and Non-Cash Interest Expense
In connection with the Purchase and Sale Agreement, the Company recognizes a liability related to the sales of future royalties under ASC 470-10, Debt and ASC 835-30, Interest - Imputation of Interest. The initial funds received by the Company pursuant to the terms of the Purchase and Sale Agreement were recorded as a liability and are accreted under the effective interest method up to the estimated amount of future royalties and milestone payments to be made under the Purchase and Sale Agreement. The issuance costs were recorded as a direct deduction to the carrying amount of the liability and are being amortized under the effective interest method over the estimated period the liability will be repaid. The Company estimates the total amount of future royalty revenue and milestone payments to be generated over the life of the Purchase and Sale Agreement, and a significant increase or decrease in these estimates could materially impact the liability balance and the related interest expense. If the timing of the receipt of royalty payments or milestones is materially different from the original estimates, the Company will prospectively adjust the effective interest and the related amortization of the liability and related issuance costs.
Warrant Liabilities
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in ASC 480, Distinguishing Liabilities from Equity (ASC 480) and ASC 815, Derivatives and Hedging (ASC 815). The assessment considers whether the warrants (i) are freestanding financial instruments pursuant to ASC 480, (ii) meet the definition of a liability pursuant to ASC 480, and (iii) meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company's own stock and whether the warrant holders could potentially require “net cash settlement” in a circumstance outside of the Company’s control, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly period end date while the warrants are outstanding.
For warrants that meet all criteria for equity classification, the warrants are required to be recorded as a component of additional paid-in capital, on the consolidated statement of stockholders’ deficit at the time of issuance. For warrants that do not meet all the criteria for equity classification, the warrants are required to be recorded at their initial fair value on the date of issuance and on each consolidated balance sheet date thereafter.
The Company’s warrant liabilities are measured at fair value using a simulation model which takes into account, as of the valuation date, factors including the current exercise price, the expected life of the warrant, the current price of the Company's common stock, the expected volatility, the risk-free interest rate for the term of the warrant and the likelihood of achieving certain future milestone events and the related impact to the price of the Company's common stock. The warrant liabilities are revalued at each reporting period and changes in fair value are recognized in other income (expense) in the consolidated statements of operations. The selection of the appropriate valuation model and the inputs and assumptions that are required to determine the valuation requires significant judgment and requires management to make estimates and assumptions that affect the reported amount of the related liability and reported amounts of the change in fair value. Actual results could differ from those estimates, and changes in these estimates are recorded when known. See Note 7 for further details.
Recently Issued Accounting Pronouncements
In December 2023, the FASB issued Accounting Standards Update ("ASU") 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures. This ASU is intended to enhance the transparency and decision usefulness of income tax disclosures primarily related to the rate reconciliation and income taxes paid information. The main provisions to the rate reconciliation disclosure require public entities to disclose, on an annual basis, specific categories in the rate reconciliation and provide additional information for reconciling items that meet a quantitative threshold. The main provisions to the income taxes paid disclosure require that all entities disclose on an annual basis: the amount of income taxes paid disaggregated by federal, state and foreign taxes and the amount of income taxes paid disaggregated by individual jurisdictions in which income taxes paid meets a quantitative threshold. This ASU also requires all entities to disclose income (loss) from continuing operations before income tax expense (benefit) disaggregated between domestic and foreign and income tax expense (benefit) from continuing operations disaggregated by federal, state and foreign. The Company adopted ASU 2023-09 on January 1, 2025 and the adoption did not have a material impact on its consolidated financial statements.
In November 2024, the FASB issued ASU 2024-03, Income Statement-Reporting Comprehensive Income-Expense Disaggregation Disclosures. This ASU requires disclosure of disaggregated income statement expense information about specific categories including purchases of inventory, employee compensation expense, depreciation and amortization in the notes to the
financial statements. This ASU is effective January 1, 2027 for annual reporting periods and January 1, 2028 for interim reporting periods. The Company is currently evaluating the anticipated impact of this ASU on its consolidated financial statements.
3. Property and Equipment, Net
Property and equipment, net consisted of the following (dollar amounts in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated
Useful Lives
(Years) |
|
June 30,
2025 |
|
|
December 31,
2024 |
|
Furniture and fixtures |
|
5 |
|
$ |
249 |
|
|
$ |
249 |
|
Machinery and equipment |
|
5 |
|
|
1,759 |
|
|
|
1,756 |
|
Computer equipment |
|
3 |
|
|
20 |
|
|
|
20 |
|
Leasehold improvements |
|
Lesser of
useful life or
remaining
lease term |
|
|
476 |
|
|
|
476 |
|
Work in process |
|
|
|
|
1,876 |
|
|
|
1,870 |
|
Total property and equipment |
|
|
|
|
4,380 |
|
|
|
4,371 |
|
Less: Accumulated depreciation |
|
|
|
|
(1,315 |
) |
|
|
(1,146 |
) |
Property and equipment, net |
|
|
|
$ |
3,065 |
|
|
$ |
3,225 |
|
4. Accrued Liabilities
Accrued liabilities consisted of the following (in thousands):
|
|
|
|
|
|
|
|
|
|
|
June 30, |
|
|
December 31, |
|
|
|
2025 |
|
|
2024 |
|
Accrued research and development |
|
$ |
451 |
|
|
$ |
545 |
|
Accrued income taxes |
|
|
692 |
|
|
|
— |
|
Accrued employee costs |
|
|
194 |
|
|
|
2,159 |
|
Accrued professional fees |
|
|
116 |
|
|
|
37 |
|
Accrued expense |
|
|
84 |
|
|
|
226 |
|
|
|
$ |
1,537 |
|
|
$ |
2,967 |
|
5. Royalty Purchase and Sale Agreement
On August 8, 2022 (the “Closing Date”), the Company, through its wholly owned subsidiary Clearside Royalty LLC, a Delaware limited liability company ("Royalty Sub"), entered into the Purchase and Sale Agreement (the "Purchased and Sale Agreement") with entities managed by HealthCare Royalty Management, LLC ("HCR"), pursuant to which Royalty Sub sold to HCR certain of its rights to receive royalty and milestone payments payable to Royalty Sub under the Arctic Vision License Agreement (as defined in Note 10), the Bausch License Agreement (as defined in Note 10), that certain License Agreement, effective as of July 3, 2019, by and between the Company and Aura Biosciences, Inc. (the “Aura License Agreement”), that certain Option and License Agreement, dated as of August 29, 2019, by and between REGENXBIO Inc. and the Company (the “REGENXBIO License Agreement”), and any and all out-license agreements following the Closing Date for, or related to XIPERE or the SCS Microinjector technology (to be used in connection with compounds or products of any third parties) delivered, in whole or in part, by means of the SCS Microinjector technology) (collectively, "Post-Closing License Agreement"), excluding, for the avoidance of doubt, any in-licensed or internally developed therapies following the Closing Date (collectively, the “Royalties”), in exchange for up to $65 million. In connection with this transaction, the Company assigned the Arctic Vision License Agreement, Bausch License Agreement, Aura License Agreement, REGENXBIO License Agreement, the Company's license agreement with Emory University and The Georgia Tech Research Corporation and related intellectual property rights to Royalty Sub. On November 1, 2023, the Company entered into the BioCryst License Agreement (as defined in Note 10). The Company’s rights to milestone payments and royalties under the BioCryst License Agreement were sold to HCR pursuant to the terms of the Purchase and Sale Agreement providing for the sale of Royalties from Post-Closing License Agreements to HCR.
Under the terms of the Purchase and Sale Agreement, Royalty Sub received an initial payment of $32.1 million, representing the $32.5 million to which the Company was entitled, net of certain of HCR's transaction-related expenses which the Company agreed to reimburse. There were additional issuance costs of $1.5 million related to the Purchase and Sale Agreement resulting in net proceeds
of $30.6 million. An additional $12.5 million (the "First Milestone Payment") was deposited by HCR in an escrow account which was released to HCR pursuant to the Letter Agreement described below.
The Purchase and Sale Agreement will automatically expire, and the payment of Royalties from the Royalty Sub to HCR will cease, when HCR has received payments of the Royalties equal to 3.4 times the aggregate amount of payments under the Purchase and Sale Agreement (the “Cap Amount”). In the event of a change in control, acquiror will have the option to make a payment to HCR of the Cap Amount, less the aggregate amount of Royalty payments made by Royalty Sub to HCR under the Purchase and Sale Agreement as a one-time payment at which time, payment of Royalties to HCR will cease. Alternatively, in the event of a change in control, the acquiror will have the option to make an initial payment of 1.0 times the aggregate amount of payments made by HCR under the Purchase and Sale Agreement as of the date of such change in control, then in that event, payment of Royalties from Royalty Sub to HCR will cease when HCR has received total Royalties payments (including the initial payment) equal to the Cap Amount. After the Purchase and Sale Agreement expires, all rights to receive the Royalties return to Royalty Sub.
On December 22, 2023, the Company, through its wholly owned subsidiary Royalty Sub, entered into the Letter Agreement with the Agent amending the Purchase and Sale Agreement. Pursuant to the terms of the Letter Agreement, Royalty Sub and Agent mutually agreed that Royalty Sub waived any and all rights to the First Milestone Payment in connection with the closing of the transactions contemplated by the Purchase and Sale Agreement and agreed to the release of the First Milestone Payment to Agent.
Issuance costs pursuant to the Purchase and Sale Agreement consisting primarily of advisory and legal fees, totaled $1.9 million including the amount of HCR's transaction-related expenses that the Company reimbursed. The effective interest rate includes cash flow projections for future royalty and milestone payments, which are sensitive to certain assumptions, including market size, market penetration and sales price, that are forward looking and could be affected by future market conditions. The Company estimates the amount and timing of expected payments based on historical experience and its expectations of future activities from its license partners, as well as current market conditions.
The following table summarizes the activity of the Purchase and Sale Agreement (in thousands):
|
|
|
|
|
Balance at December 31, 2023 |
|
$ |
41,988 |
|
Non-cash interest expense |
|
|
9,779 |
|
Balance at December 31, 2024 |
|
|
51,767 |
|
Payments |
|
|
(1,500 |
) |
Non-cash interest expense |
|
|
5,435 |
|
Balance at June 30, 2025 |
|
$ |
55,702 |
|
|
|
|
|
Effective interest rate |
|
|
21.8 |
% |
6. Common Stock
At the Company's 2025 annual meeting of stockholders held on May 30, 2025, the Company's stockholders approved an amendment to the Company's Amended and Restated Certificate of Incorporation to increase the Company's authorized number of shares of common stock from 200,000,000 to 400,000,000. As of June 30, 2025, the Company was authorized to issue 400,000,000 shares of common stock, $0.001 par value. As of June 30, 2025 and December 31, 2024, there were 78,495,760 and 76,578,383 shares of common stock outstanding, respectively.
7. Common Stock Warrants
In September 2016, in connection with a loan agreement, the Company issued warrants to purchase up to 29,796 shares of common stock at a price per share of $10.74. The warrants are fully exercisable and expire in September 2026, or earlier upon the occurrence of specified mergers or acquisitions of the Company. The warrants were recorded in equity at the time of issuance and as of June 30, 2025, had a weighted average remaining life of 1.25 years.
On February 6, 2024, the Company entered into a securities purchase agreement with institutional investors and an existing stockholder, pursuant to which the Company issued and sold, in a registered direct offering (i) an aggregate of 11,111,111 shares (the "Shares") of its common stock; and (ii) warrants to purchase up to 11,111,111 shares of common stock (the "Warrants").
The combined purchase price of each Share and accompanying Warrant was $1.35. The exercise price for the Warrants is $1.62 per share. The Warrants are currently exercisable and will expire on August 9, 2029. The Company recorded the initial fair value of the Warrants of $10.3 million as warrant liabilities and $4.7 million attributable to common stock as additional paid in capital in the consolidated balance sheets. The issuance costs were allocated among the Warrants and common stock consistent with the allocation between amounts recorded as warrant liabilities and common stock. The issuance costs allocated to the Warrants as well as the change
in the fair value of the Warrants during the period are recorded in other income, net in the consolidated statements of operations. The issuance costs allocated to common stock were recorded as a reduction to additional paid in capital.
The following table summarizes the change in fair value of the warrant liabilities (in thousands):
|
|
|
|
|
Fair value of warrants at issuance February 9, 2024 |
|
$ |
10,327 |
|
Change in fair value during the period |
|
|
(3,635 |
) |
Fair value of warrants at December 31, 2024 |
|
|
6,692 |
|
Change in fair value during the period |
|
|
(1,927 |
) |
Fair value of warrants at June 30, 2025 |
|
$ |
4,765 |
|
The following table summarizes certain key inputs for the valuation of the Warrants at June 30 2025:
|
|
|
|
|
|
Common stock price |
|
$ |
0.80 |
|
|
Exercise price per share |
|
$ |
1.62 |
|
|
Expected volatility |
|
|
67.00 |
|
% |
Risk-free interest rate |
|
|
3.67 |
|
% |
Contractual term (in years) |
|
|
4.11 |
|
|
Expected dividend yield |
|
|
— |
|
% |
As described in Note 2, the measurement of the warrant liabilities is impacted by the likelihood of achieving certain future milestone events and the related impact to the Company’s stock price. In determining the likelihood of achieving certain future milestone events, the Company considers its current financial position and ability to fund future clinical activities as described in Note 1. The Company utilizes publicly available information from external parties to assess the related impact to the Company’s stock price from the success of these activities.
As described in Note 1, the Company is exploring strategic alternatives. Following the decision to explore such alternatives and related announcement in July 2025, the Company experienced a significant decline in its traded stock price. This decline, coupled with the increased uncertainty in the Company’s future financial viability, has led to a further decline in the fair value of the Company's warrant liabilities subsequent to June 30, 2025.
8. Share-Based Compensation
Share-based compensation is accounted for in accordance with the provisions of ASC 718, Compensation-Stock Compensation.
Stock Options
The Company has granted stock option awards to employees, directors and consultants from its 2011 Stock Incentive Plan (the “2011 Plan”) and its 2016 Equity Incentive Plan (the “2016 Plan”). The estimated fair value of options granted is determined as of the date of grant using the Black-Scholes option pricing model. The resulting fair value is recognized ratably over the requisite service period, which is generally the vesting period of the awards.
Share-based compensation expense for options granted under the 2016 Plan is reflected in the consolidated statements of operations as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
Research and development |
|
$ |
256 |
|
|
$ |
384 |
|
|
$ |
502 |
|
|
$ |
804 |
|
General and administrative |
|
|
365 |
|
|
|
480 |
|
|
|
734 |
|
|
|
862 |
|
Total |
|
$ |
621 |
|
|
$ |
864 |
|
|
$ |
1,236 |
|
|
$ |
1,666 |
|
The following table summarizes the activity related to stock options granted under the 2011 Plan and the 2016 Plan during the six months ended June 30, 2025:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted |
|
|
|
Number of |
|
|
Average |
|
|
|
Shares |
|
|
Exercise Price |
|
Options outstanding at December 31, 2024 |
|
|
12,644,605 |
|
|
$ |
2.46 |
|
Granted |
|
|
2,260,584 |
|
|
|
0.97 |
|
Forfeited |
|
|
(575,000 |
) |
|
|
1.29 |
|
Options outstanding at June 30, 2025 |
|
|
14,330,189 |
|
|
|
2.28 |
|
|
|
|
|
|
|
|
Options exercisable at December 31, 2024 |
|
|
7,643,102 |
|
|
|
3.19 |
|
|
|
|
|
|
|
|
Options exercisable at June 30, 2025 |
|
|
9,168,262 |
|
|
|
2.89 |
|
As of June 30, 2025, the Company had $4.0 million of unrecognized compensation expense related to unvested stock options, which is expected to be recognized over a weighted average period of 2.6 years.
Restricted Stock Units
The Company has granted restricted stock units (“RSUs”) to employees and consultants under the 2016 Plan. The shares underlying the RSU awards have vesting terms of four years from the date of grant subject to the recipient's continuous service and subject to accelerated vesting in specified circumstances. The fair value of the RSUs granted is measured based on the market value of the Company’s common stock on the date of grant and is recognized ratably over the requisite service period, which is generally the vesting period of the awards.
The total share-based compensation expense related to RSUs is reflected in the consolidated statements of operations as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
Research and development |
|
$ |
33 |
|
|
$ |
118 |
|
|
$ |
83 |
|
|
$ |
237 |
|
General and administrative |
|
|
36 |
|
|
|
135 |
|
|
|
91 |
|
|
|
273 |
|
Total |
|
$ |
69 |
|
|
$ |
253 |
|
|
$ |
174 |
|
|
$ |
510 |
|
The following table summarizes the activity related to RSUs during the six months ended June 30, 2025:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted Average |
|
|
|
Number of |
|
|
Grant Date |
|
|
|
Shares |
|
|
Fair Value |
|
Non-vested RSUs outstanding at December 31, 2024 |
|
|
437,305 |
|
|
$ |
2.96 |
|
Vested |
|
|
(310,797 |
) |
|
|
3.27 |
|
Non-vested RSUs outstanding at June 30, 2025 |
|
|
126,508 |
|
|
|
2.19 |
|
As of June 30, 2025, the Company had $0.2 million of unrecognized compensation expense related to the RSUs which is expected to be recognized over a weighted average period of 0.6 years.
Employee Stock Purchase Plan
The 2016 Employee Stock Purchase Plan (the “2016 ESPP”) became effective on June 1, 2016. The 2016 ESPP is considered a compensatory plan and the fair value of the discount and the look-back period are estimated using the Black-Scholes option pricing model and expense is recognized over the six-month withholding period prior to the purchase date.
The share-based compensation expense recognized for the 2016 ESPP is reflected in the consolidated statements of operations as follows (in thousands):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
Research and development |
|
$ |
3 |
|
|
$ |
2 |
|
|
$ |
4 |
|
|
$ |
5 |
|
General and administrative |
|
|
1 |
|
|
|
1 |
|
|
|
1 |
|
|
|
1 |
|
Total |
|
$ |
4 |
|
|
$ |
3 |
|
|
$ |
5 |
|
|
$ |
6 |
|
During the six months ended June 30, 2025, the Company issued 16,222 shares of common stock purchased under the 2016 ESPP.
9. Commitments and Contingencies
Lease Commitment Summary
In November 2022, the Company signed an amended office lease agreement to lease approximately 14,000 square feet of office space in Alpharetta, Georgia for its corporate headquarters. The amended office lease agreement is for a four year term with a renewal option for an additional 38 months. Rental payments are $30,747 per month subject to an increase of 3% per year. Rent expense under this lease is recognized on a straight-line basis over the term of the lease. In addition, the office lease agreement requires payment of the pro-rata share of the annual operating expenses associated with the premises.
The Company recognizes a right-of-use asset for the right to use the underlying asset for the lease term, and a lease liability, which represents the present value of the Company’s obligation to make payments over the lease term. The renewal option is not included in the calculation of the right-of-use asset and the lease liabilities as the Company has not yet determined if the Alpharetta, Georgia lease will be renewed.
Equipment leases with an initial term of 12 months or less are not recorded with operating lease liabilities. The Company recognizes expense for these leases on a straight-line basis over the lease term. The equipment leases were deemed to be immaterial.
Georgia Tech License Agreement
As described in Note 1, the Company entered into a fourth amendment to the Georgia Tech License Agreement pursuant to which the parties agreed to revised Maintenance Fee payments in exchange for a reduction to the contractual Sublicense Percentage owed by the Company on certain fees and other payments it may receive from future sublicensing activities. The Company paid the $0.3 million Maintenance Fee for 2023 in February 2024 and the $0.3 million Maintenance Fee for 2024 in October 2024. The remaining annual Maintenance Fee payments are due on October 1st of each year from 2025 through 2028, as shown in the table below (in thousands).
|
|
|
|
|
Year Ending December 31, |
|
Amount |
|
2025 |
|
$ |
250 |
|
2026 |
|
|
350 |
|
2027 |
|
|
400 |
|
2028 |
|
|
500 |
|
Total |
|
$ |
1,500 |
|
Contract Service Providers
In the course of the Company’s normal business operations, it has agreements with contract service providers to assist in the performance of its research and development, clinical research and manufacturing. Substantially all of these contracts are on an as needed basis.
10. License and Other Agreements
Bausch + Lomb
On October 22, 2019, the Company entered into a License Agreement (as amended, the "Bausch License Agreement") with Bausch + Lomb (“Bausch”). Pursuant to the Bausch License Agreement, the Company has granted an exclusive license to Bausch to develop, manufacture, distribute, promote, market and commercialize XIPERE using the Company’s proprietary SCS Microinjector (the “Device”), as well as specified other steroids, corticosteroids and NSAIDs in combination with the Device (together with
XIPERE, the “Products”), subject to specified exceptions, in the United States and Canada (the “Territory”) for the treatment of ophthalmology indications, including non-infectious uveitis.
Pursuant to the Bausch License Agreement, Bausch paid the Company an aggregate of $20.0 million in upfront and milestone payments. In addition, Bausch has agreed to pay up to an aggregate of $55.0 million in additional milestone payments upon the achievement of (i) specified regulatory approvals for specified additional indications of XIPERE and (ii) specified levels of annual net sales (as defined in the Bausch License Agreement). Further, during the applicable royalty term, the Company will also be entitled to receive tiered royalties at increasing percentages, from the high-teens to twenty percent, based on XIPERE achieving certain annual net sales thresholds in the Territory, in each case subject to reductions in specified circumstances; provided that the Company will not receive any royalties on the first $45.0 million of cumulative net sales of all products in the Territory. Bausch launched XIPERE in the United States in the first quarter of 2022. The Company's rights to these royalties and milestone payments have been sold pursuant to the terms and conditions of the Purchase and Sale Agreement described in Note 5.
The Company was responsible for all development expenses for XIPERE in the Territory until the Company's New Drug Application ("NDA") was approved by the U.S. Food and Drug Administration ("FDA"), subject to specified exceptions, as well as manufacturing costs in connection with the NDA. The Company was also responsible for all clinical and development expenses conducted to satisfy the FDA’s requests in the complete response letter issued on October 18, 2019 related to the NDA. Following FDA approval of XIPERE, which occurred in October 2021, Bausch is responsible for all such expenses.
Arctic Vision (Hong Kong) Limited
On March 10, 2020, the Company entered into a License Agreement (the “Arctic License Agreement”) with Arctic Vision (Hong Kong) Limited (“Arctic Vision”). Pursuant to the Arctic License Agreement, the Company has granted an exclusive license to Arctic Vision to develop, distribute, promote, market and commercialize XIPERE, subject to specified exceptions, in China, Hong Kong, Macau, Taiwan and South Korea (the “Arctic Territory”). Under the terms of the Arctic License Agreement, neither party may commercialize XIPERE in the other party’s territory. Arctic Vision has agreed to use commercially reasonable efforts to pursue the development and commercialization of XIPERE for indications associated with uveitis in the Arctic Territory. In addition, upon receipt of the Company’s consent, Arctic Vision will have the right, but not the obligation, to develop and commercialize XIPERE for additional indications in the Arctic Territory.
Pursuant to the Arctic License Agreement, Arctic Vision has paid the Company an aggregate of $10.5 million in upfront and milestone payments. In addition, Arctic Vision has agreed to pay the Company up to $22.0 million in development and sales milestones. Further, during the applicable royalty term, the Company will also be entitled to receive tiered royalties of ten to twelve percent of net sales based on achieving certain annual net sales thresholds in the Arctic Territory, subject to customary reductions, payable on a product-by-product and country-by-country basis, commencing at launch in such country and lasting until the latest of (i) the date that all valid claims within the licensed patent rights covering XIPERE have expired, (ii) the date of the loss of marketing or regulatory exclusivity of XIPERE in a given country, or (iii) ten years from the first commercial sale of XIPERE in a given country. The Company's rights to these royalties and milestone payments have been sold pursuant to the terms and conditions of the Purchase and Sale Agreement described in Note 5.
In August 2021, the Company entered into an amendment to the Arctic License Agreement to expand the territories covered by the license to include India and the ASEAN Countries (Brunei, Cambodia, Indonesia, Laos, Malaysia, Myanmar, the Philippines, Singapore, Thailand, and Vietnam). In September 2021, the Company entered into a second amendment to the Arctic Vision License Agreement to expand the Arctic Territory to include Australia and New Zealand. The Company received an aggregate of $3.0 million in consideration for the expansion of the Arctic Territory.
BioCryst Pharmaceuticals, Inc.
On November 1, 2023, the Company entered into the BioCryst License Agreement pursuant to which the Company granted BioCryst an exclusive, worldwide and sublicensable license to the Company’s SCS Microinjector for the delivery of BioCryst’s proprietary plasma kallikrein inhibitor known as avoralstat for the treatment and prevention of diabetic macular edema.
The Company received an upfront license fee payment of $5.0 million in connection with signing of the BioCryst License Agreement. In addition, the Company is eligible to receive up to an additional $30.0 million in clinical and regulatory milestone payments, and up to a total of $47.5 million in a series of post-approval sales-based milestone payments based on the achievement of annual global net product sales milestones up to $2.0 billion. Further, during the royalty term, BioCryst has also agreed to pay the Company tiered mid-single digit royalties on annual global net product sales, with the highest royalty rate applied to sales over $1.5 billion, subject to reductions in specified circumstances. The Company's rights to these royalties and milestone payments have been sold pursuant to the terms and conditions of the Purchase and Sale Agreement described in Note 5.
BioCryst will be responsible for all development, regulatory and commercialization activities for avoralstat. The Company is responsible for supplying SCS Microinjectors to meet BioCryst’s reasonable needs.
Other
The Company periodically enters into short-term agreements with other customers to evaluate the potential use of its proprietary SCS Microinjector with third-party product candidates for the treatment of various diseases. Funds received from these agreements are recognized as revenue over the term of the agreement.
11. Fair Value Measurements
The Company’s material financial instruments at June 30, 2025 and December 31, 2024 consisted of cash and cash equivalents. The fair values of cash and cash equivalents, other current assets and accounts payable approximate their respective carrying values due to the short term nature of these instruments and are classified as Level 1 in the fair value hierarchy. The fair value of the warrant liabilities (see Note 7) require significant unobservable inputs and is classified as Level 3 in the fair value hierarchy.
There were no transfers between Levels 1, 2 and 3 during the six months ended June 30, 2025 and the year ended December 31, 2024.
12. Related Party Transactions
A member of the Company's Board of Directors was the chief executive officer of a company that is a vendor of the Company until January 2025. As of December 31, 2024, the Company has recorded $0.5 million in accounts payable and $0.3 million in accrued expense with this vendor in the consolidated balance sheet. The Company has recorded $0.2 million and $0.5 million for the three and six months ended June 30, 2024, respectively, of expense related to this vendor in the consolidated statement of operations.
The chair of the board of directors of BioCryst also serves on the Company’s Board of Directors. The Company has recorded $0.3 million and $81,000 for the three months ended June 30, 2025 and 2024, respectively, and $0.3 million and $0.2 million for the six months ended in June 30 2025 and 2024, respectively, in license and other revenue in the consolidated statements of operations related to the BioCryst License Agreement.
A member of the Company's Board of Directors is the chief financial officer of Aura Biosciences. The Company has recorded $0.1 million in license and other revenue in the consolidated statement of operations related to the Aura License Agreement for each of the three and six month periods ending June 30, 2025.
13. Net Loss Per Share
Basic net loss per share is calculated by dividing the net loss by the weighted average number of shares of common stock outstanding for the period, without consideration of the dilutive effect of potential common stock equivalents. Diluted net loss per share gives effect to all dilutive potential shares of common stock outstanding during this period. For all periods presented, the Company’s potential common stock equivalents, which included stock options, restricted stock units and common stock warrants, have been excluded from the computation of diluted net loss per share as their inclusion would have the effect of reducing the net loss per share. Therefore, the denominator used to calculate both basic and diluted net loss per share is the same in all periods presented.
The Company’s potential common stock equivalents that have been excluded from the computation of diluted net loss per share for all periods presented because of their antidilutive effect consisted of the following:
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
Outstanding stock options |
|
|
14,330,189 |
|
|
|
12,767,330 |
|
Non-vested restricted stock units |
|
|
126,508 |
|
|
|
437,305 |
|
Common stock warrants |
|
|
11,140,907 |
|
|
|
11,140,907 |
|
|
|
|
25,597,604 |
|
|
|
24,345,542 |
|
14. Subsequent Event
On July 17, 2025, the Company announced plans to explore a full range of strategic alternatives to advance its SCS platform and drug development pipeline to maximize stockholder value. The Company has retained Piper Sandler, a leading investment bank with substantial experience in the biotechnology industry, to support it with the strategic evaluation process. Strategic alternatives under consideration include the sale, license, monetization and/or divestiture of one or more of the Company’s assets and technologies, collaboration, partnership, merger, acquisition, joint ventures, or other strategic transactions. If a strategic alternative is not available, the Company will be required to take additional actions to fund the Company’s operations, or it may be forced to file for bankruptcy or wind down its operations. In connection with such process and in order to extend the Company’s resources, the Company
implemented a plan pursuant to which all Company employees had their employment with the Company terminated and transitioned into consulting roles with the Company. The reduction in force is expected to be completed during the third quarter of 2025.
The Company estimates that it will incur charges of approximately $2.6 million for severance and other employee termination-related costs in the third quarter of 2025. The estimated charges that the Company expects to incur are subject to a number of assumptions, and actual results may differ materially from these estimates. The Company may also incur additional costs not currently contemplated due to events that may occur as a result of, or that are associated with, its workforce reduction.
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
Certain statements contained in this Quarterly Report on Form 10-Q may constitute forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. The words or phrases “would be,” “will allow,” “intends to,” “will likely result,” “are expected to,” “will continue,” “is anticipated,” “estimate,” “project,” or similar expressions, or the negative of such words or phrases, are intended to identify “forward-looking statements.” We have based these forward-looking statements on our current expectations and projections about future events. Because such statements include risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Factors that could cause or contribute to these differences include those below and elsewhere in this Quarterly Report on Form 10-Q and our other filings with the Securities and Exchange Commission, or SEC, under the heading “Risk Factors”. Statements made herein are as of the date of the filing of this Form 10-Q with the SEC and should not be relied upon as of any subsequent date. Unless otherwise required by applicable law, we do not undertake, and we specifically disclaim, any obligation to update any forward-looking statements to reflect occurrences, developments, unanticipated events or circumstances after the date of such statement.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our unaudited consolidated financial statements and related notes that appear in Item 1 of this Quarterly Report on Form 10-Q and with our audited consolidated financial statements and related notes for the year ended December 31, 2024 appearing in our Annual Report on Form 10-K filed with the SEC on March 27, 2025.
Overview
We are a biopharmaceutical company focused on revolutionizing the delivery of therapies to the back of the eye through the suprachoroidal space, or SCS®. Our novel SCS injection platform, utilizing our proprietary SCS Microinjector®, enables an in-office, repeatable, non-surgical procedure for the targeted and compartmentalized delivery of a wide variety of therapies to the macula, retina or choroid to potentially preserve and improve vision in patients with sight-threatening eye diseases. Our SCS injection platform can be used in conjunction with existing drugs designed for delivery to the SCS, novel therapies and future therapeutic innovations. We believe our proprietary suprachoroidal administration platform has the potential to become a standard for delivery of therapies intended to treat chorioretinal diseases.
We are leveraging our SCS injection platform with an internal research and development pipeline targeting retinal diseases and through external collaborations with other companies. We have our own pipeline of small molecule product candidates for administration via our SCS Microinjector, and we also strategically partner with companies developing other ophthalmic therapeutic innovations to be administered using our SCS injection technology. Our first product, XIPERE® (triamcinolone acetonide injectable suspension) for suprachoroidal use, was approved by the U.S. Food and Drug Administration, or the FDA, in October 2021. Approval of XIPERE was a significant milestone for us as it is the first approved therapeutic delivered into the SCS, the first commercial product developed by us and the first therapy for macular edema associated with uveitis.
We believe that we have a broad therapeutic platform for developing product candidates to treat serious eye diseases.
In connection with our July 2025 announcement of plans to explore strategic alternatives and in order to extend resources, we paused all internal research and development programs during the pendency of this process. The current development status of our pipeline of internal product candidates and external collaborations is summarized in the charts below:
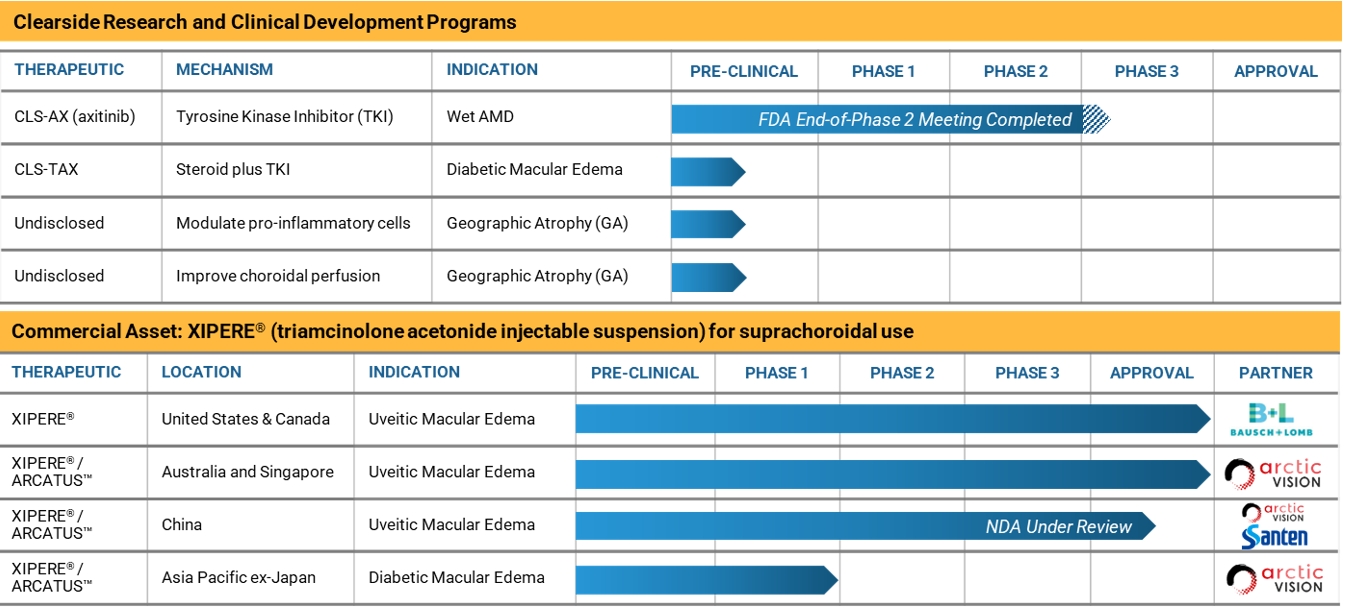
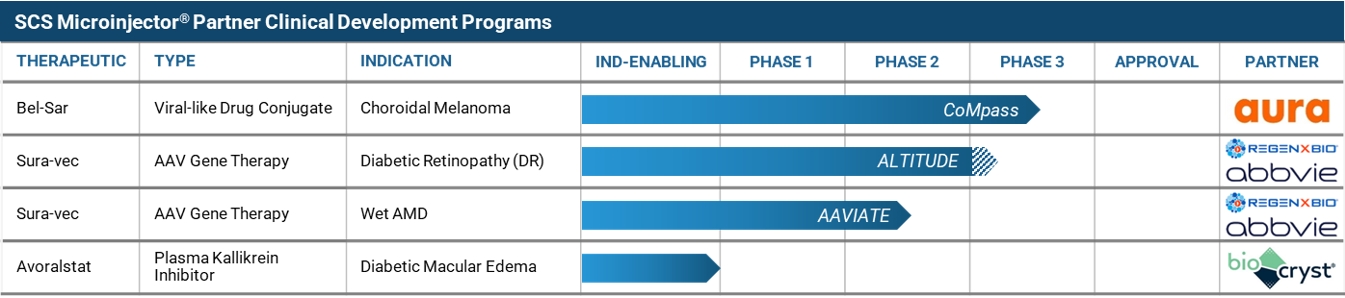
Clinical Development Pipeline
CLS-AX (axitinib injectable suspension)
CLS-AX, our most advanced product candidate, is our proprietary suspension of the tyrosine kinase inhibitor, or TKI, axitinib for suprachoroidal injection delivered via our SCS Microinjector. CLS-AX is a Phase 3 ready asset for administration to the SCS as a long-acting therapy for neovascular age-related macular degeneration, or wet AMD, a retinal degenerative disease that causes a progressive loss of central vision.
ODYSSEY Phase 2b Clinical Trial
Based on the results from our OASIS trial, we conducted ODYSSEY, a randomized, active-controlled, double-masked, parallel-group, multicenter, 36-week, Phase 2b clinical trial of CLS-AX in participants with wet AMD previously treated with intravitreal anti-vascular, or VEGF, which is the standard of care therapy for the treatment of wet AMD. A total of 60 participants were treated for 36 weeks and randomized to either CLS-AX (1 mg) or aflibercept (2 mg) with a 2:1 randomization schedule (40 participants in CLS-AX arm and 20 participants in aflibercept arm). CLS-AX was administered by suprachoroidal injection via our SCS Microinjector, and aflibercept was administered via intravitreal injection. Participants in the trial were determined to have active disease with a median duration of wet AMD diagnosis of 9.9 months. Eligible participants underwent diagnostic imaging at their screening visit, followed by masked reading center confirmation of persistent active disease.
In October 2024, we announced positive topline results from the ODYSSEY trial. The ODYSSEY trial achieved its primary and secondary outcomes including the mean change from baseline in BCVA, changes from baseline in visual function and ocular anatomy, the need for supplemental treatment, treatment burden as measured by total injections over the trial duration, and safety measures.
Participants in the trial maintained stable BCVA throughout the trial as measured by the mean change in BCVA from baseline to week 36. In addition, participants maintained stable central subfield retinal thickness, or CSRT, throughout the trial as measured by the mean change in CSRT from baseline to week 36, as confirmed by the independent reading center.
In terms of durability, after receiving the baseline CLS-AX dose, 100% (40/40) of participants went 3 months without receiving any additional treatment, 90% (35/39) of participants went 4 months without receiving any additional treatment, 81% (30/37) of participants went five months without receiving any additional treatment and 67% (26/39) of participants went 6 months without receiving any additional treatment before mandatory re-dosing at week 24. If intervention were strictly based on the disease activity assessment criteria, 100% (40/40) of participants would have gone 3 months without receiving any additional treatment, 95% (37/39) of CLS-AX participants would have gone 4 months without receiving any additional treatment, 87% (32/37) of participants would have gone five months without receiving any additional treatment and 79% (30/38) of participants would have gone six months without receiving any additional treatment before mandatory re-dosing at week 24.
Additionally, we observed reduced injection frequency by 84% in the 24 weeks after baseline compared to the average monthly injections in the 24 weeks prior to screening.
CLS-AX was well-tolerated with a safety profile through 36 weeks that included mandatory re-dosing of CLS-AX at week 24. There were no reported ocular serious adverse events, or SAEs, and no treatment or injection procedure-related SAEs. All ocular adverse events were considered clinically mild in both groups. We observed similar discontinuation rates in the CLS-AX and aflibercept arms.
CLS-AX Phase 3
We conducted an End-of-Phase 2 meeting with the FDA in the first quarter of 2025 and gained alignment on the essential components of a potential Phase 3 program, which would include two pivotal, non-inferiority Phase 3 trials with aflibercept 2 mg as a comparator.
Preclinical
We have evaluated various small molecules that may be utilized as potential treatment options for back of the eye diseases utilizing our SCS Microinjector for delivery in the suprachoroidal space. Our preclinical pipeline includes two small molecules evaluated using in vivo models for the potential treatment of geographic atrophy, or GA, CLS-AX for the treatment of diabetic retinopathy and the combination of CLS-AX and a steroid, triamcinolone acetonide, for the treatment of diabetic macular edema.
External Collaborations Pipeline
In order to expand the global reach of our suprachoroidal injection platform, we have strategically partnered some of our assets for development and/or commercialization and intend to continue partnering our assets. By entering into these partnerships, we have been able to expand the use of our suprachoroidal injection platform to other indications and geographies globally. We currently have collaborations with Bausch + Lomb, Arctic Vision, REGENXBIO, Inc., Aura Biosciences and BioCryst Pharmaceuticals, Inc.
Commercial Product
XIPERE® (triamcinolone acetonide injectable suspension) for suprachoroidal use, was approved by the FDA in October 2021. XIPERE is the first approved therapeutic delivered into the SCS, the first commercial product developed by us and the first therapy for macular edema associated with uveitis. XIPERE commercialization rights are licensed to Bausch + Lomb in the United States and Canada and Arctic Vision in the Asia Pacific region, not including Japan. In July 2025, Health Canada granted approval for XIPERE for the treatment of uveitic macular edema.
Operating Outlook and Strategic Review
We have incurred net losses since our inception. In recent years, our operations have consisted primarily of conducting preclinical studies and clinical trials, raising capital and undertaking other research and development initiatives. To date, we have primarily generated revenue through upfront payments and milestone payments related to license agreements and from collaboration agreements, and we have primarily financed our operations through public offerings and private placements of our equity securities, issuance of warrants, issuances of convertible promissory notes and loan agreements. As of June 30, 2025, we had an accumulated deficit of $368.0 million. We recorded net losses of $4.5 million and $7.6 million for the three months ended June 30, 2025 and 2024, respectively, and net losses of $12.7 million and $19.4 million for the six months ended June 30, 2025 and 2024, respectively.
In July 2025, we announced plans to explore a full range of strategic alternatives to advance our SCS platform and drug development pipeline to maximize stockholder value. We have retained Piper Sandler, a leading investment bank with substantial experience in the biotechnology industry, to support us with the strategic evaluation process. Strategic alternatives under consideration
include the sale, license, monetization and/or divestiture of one or more of our assets and technologies, collaboration, partnership, merger, acquisition, joint ventures or other strategic transactions. If a strategic alternative is not available, we will be required to take additional actions to fund our operations, or we may be forced to file for bankruptcy or wind down our operations.
Based on our current plans and forecasted expenses and our cash and cash equivalents, we do not believe we will be able to fund our operations for the next 12 months from the date of this filing. These factors raise substantial doubt regarding our ability to continue as a going concern. Our consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might result should we be unable to continue as a going concern.
Macroeconomic Conditions
Unfavorable conditions in the economy in the United States and abroad may negatively affect the growth of our business and our results of operations. For example, macroeconomic events, rising inflation, the U.S. Federal Reserve raising interest rates, and conflicts in Ukraine, Russia and the Middle East have led to economic uncertainty globally. The effect of macroeconomic conditions may not be fully reflected in our results of operations until future periods. If, however, economic uncertainty increases or the global economy worsens, our business, financial condition and results of operations may be harmed. For further discussion of the potential impacts of macroeconomic events on our business, financial condition, and operating results, see “Risk Factors” included in Part I, Item 1A of the Annual Report on Form 10-K filed with the SEC on March 27, 2025.
Recent Developments
Strategic Review and Reduction in Force
As discussed in more detail under “—Operating Outlook and Strategic Review” above, in July 2025, we announced plans to explore a full range of strategic alternatives to advance our SCS platform and drug development pipeline to maximize stockholder value. In connection with such process, and in order to extend our resources, we implemented a plan pursuant to which we terminated the employment of all employees and transitioned such persons into consulting roles. We estimate that we will incur charges of approximately $2.6 million for severance and other employee termination-related costs in the third quarter of 2025. The estimated charges that we expect to incur are subject to a number of assumptions, and actual results may differ materially from these estimates. We may also incur additional costs not currently contemplated due to events that may occur as a result of, or that are associated with, our workforce reduction. The reduction in force is expected to be completed during the third quarter of 2025.
Minimum Bid Price Requirement
On February 7, 2025, we received a letter from Nasdaq, notifying us that, from December 18, 2024 to February 4, 2025, the closing bid price for our common stock was below $1.00. In accordance with Nasdaq Listing Rule 5810(c)(3)(A) we were provided an initial period of 180 calendar days, or until August 6, 2025, to regain compliance with Nasdaq’s bid price requirement (the “Minimum Bid Price Requirement”). We did not regain compliance by August 6, 2025, and as a result we expect that Nasdaq will provide notice that our common stock will be subject to delisting. We intend to appeal any delisting determination to a Nasdaq hearings panel and our common stock will remain listed on the Nasdaq Global Market until the completion of the appeal process, during which time we may regain compliance the Minimum Bid Price Requirement if our minimum bid price of listed securities closes at $1.00 per share or more for a minimum of 10 consecutive business days. In addition, on August 4, 2025, we filed a proxy statement seeking approval by our stockholders to effect a reverse split of our outstanding common stock at a ratio ranging from 1-for-5 and 1-for-15 (the “Reverse Stock Split”). If approved by stockholders, this Reverse Stock Split Proposal would permit (but not require) our board of directors to effect a reverse stock split of our common stock at any time before August 29, 2026, by a ratio of not less than 1-for-5 and not more than 1-for-15, inclusive, and a corresponding proportionate reduction in the total number of authorized shares of the Company’s common stock (the “Authorized Shares Reduction”), with such ratio to be determined by the our board of directors in its sole discretion. Our board of directors’ primary objective in proposing the Reverse Stock Split and Authorized Shares Reduction is to raise the per share trading price of our common stock. Our board of directors believes that the Reverse Stock Split and Authorized Shares Reduction will result in a higher per share trading price, which is intended to enable us to maintain the listing of our common stock on The Nasdaq Global Market and generate greater investor interest in the Company.
Components of Operating Results
License and Other Revenue
We have not generated any revenue from the sale of XIPERE and we do not expect to generate any other product revenue unless or until we obtain regulatory approval for and commercialize our other product candidates, either on our own or with a third party. The revenue received under the Bausch license agreement, as well as other certain payments from our licensees, will be recorded as non-cash revenue until we have fulfilled our obligations under the Purchase and Sale Agreement. Our revenue in recent years has been generated primarily from our license agreements. We are seeking to enter into additional licenses and other agreements with third parties to evaluate the potential use of our proprietary SCS Microinjector with the third party’s product candidates for the treatment of
various eye diseases. These agreements may include payments to us for technology access, upfront license payments, regulatory and commercial milestone payments and royalties.
Costs of Goods Sold
Cost of goods sold are related to the sales of our SCS Microinjector kits to our licensees for approved products.
Research and Development
Research and development expenses have historically consisted primarily of costs incurred for the research and development of our preclinical and clinical product candidates, which include:
•employee-related expenses, including salaries, benefits, travel and share-based compensation expense for research and development personnel;
•expenses incurred under agreements with contract research organizations, or CROs, as well as contract manufacturing organizations and consultants that conduct clinical trials and preclinical studies;
•costs associated with preclinical activities and clinical trials;
•costs associated with submitting regulatory approval applications for our product candidates;
•costs associated with training physicians on the suprachoroidal injection procedure and educating and providing them with appropriate product candidate information;
•costs associated with technology and intellectual property licenses;
•costs for our research and development facility; and
•depreciation expense for assets used in research and development activities.
We expense research and development costs to operations as incurred. These costs include preclinical activities, such as manufacturing and stability and toxicology studies, that are supportive of a product candidate itself. In addition, there are expenses related to clinical trials and similar activities for each program, including costs associated with CROs. Clinical costs are recognized based on the terms of underlying agreements, as well as an evaluation of the progress to completion of specific tasks using data such as participant enrollment, clinical site activations and additional information provided to us by our vendors about their actual costs incurred. Expenses related to activities that support more than one development program or activity, such as salaries, share-based compensation and depreciation, are not classified as direct preclinical costs or clinical costs and are separately classified as unallocated.
The following table shows our research and development expenses by program for the three and six months ended June 30, 2025 and 2024 (in thousands).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
|
2025 |
|
|
2024 |
|
CLS-AX (wet AMD program) |
|
$ |
200 |
|
|
$ |
1,763 |
|
|
$ |
664 |
|
|
$ |
4,021 |
|
GA (geographic atrophy) |
|
|
90 |
|
|
|
— |
|
|
|
465 |
|
|
|
— |
|
Total |
|
|
290 |
|
|
|
1,763 |
|
|
|
1,129 |
|
|
|
4,021 |
|
Unallocated |
|
|
1,294 |
|
|
|
2,840 |
|
|
|
4,918 |
|
|
|
6,197 |
|
Total research and development expense |
|
$ |
1,584 |
|
|
$ |
4,603 |
|
|
$ |
6,047 |
|
|
$ |
10,218 |
|
Our expenses related to clinical trials are based on estimates of participant enrollment and related expenses at clinical investigator sites as well as estimates for the services received and efforts expended under contracts with research institutions, consultants and CROs that conduct and manage clinical trials on our behalf. We generally accrue expenses related to clinical trials based on contracted amounts applied to the level of participant enrollment and activity according to the protocol. If future timelines or contracts are modified based upon changes in the clinical trial protocol or scope of work to be performed, we would modify our estimates of accrued expenses accordingly on a prospective basis.
As discussed in more detail under “—Operating Outlook and Strategic Review” above, in July 2025, we announced plans to explore a full range of strategic alternatives to advance our SCS platform and drug development pipeline to maximize stockholder value. We paused all internal research and development programs during the pendency of this process. Accordingly, we expect research and development expenses will continue to decrease in future periods relative to the corresponding periods of 2024.
General and Administrative
General and administrative expenses consist primarily of salaries and other related costs, including share-based compensation, for personnel in executive, finance and administrative functions. General and administrative costs historically included commercial pre-launch preparations for XIPERE, and also include facility related costs not otherwise included in research and development expenses, as well as professional fees for legal, patent, consulting, and accounting and audit services.
Interest Income
Interest income consists of the accrued interest and interest income earned on our cash and cash equivalents.
Other Income, Net
Other income, net consists of expenses allocated to the warrants issued in connection with our registered direct offering in February 2024 and the change in fair value of the warrants during the period.
Non-cash Interest Expense on Liability Related to the Sales of Future Royalties
Non-cash interest expense on liability related to the sales of future royalties consists of imputed interest on the carrying value of the liability and the amortization of the related issuance costs.
Income Tax Expense
Income tax expense consists of taxes incurred in foreign jurisdictions.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles or U.S. GAAP. The preparation of these consolidated financial statements requires us to make estimates, judgments and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities as of the dates of the consolidated balance sheets and the reported amounts of expenses during the reporting periods. In accordance with U.S. GAAP, we evaluate our estimates and judgments on an ongoing basis. Significant estimates include certain assumptions used in royalty financing obligation, certain assumptions used in the valuation of warrant liabilities and certain assumptions used to estimate research and development expenses, including clinical trials. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
We define our critical accounting policies, in accordance with U.S. GAAP, as those that require us to make subjective estimates and judgments about matters that are uncertain and are likely to have a material impact on our financial condition and results of operations, as well as the specific manner in which we apply those principles. During the six months ended June 30, 2025, there were no significant changes to our critical accounting policies disclosed in our audited consolidated financial statements for the year ended December 31, 2024, which are included in our Annual Report on Form 10-K, as filed with the SEC on March 27, 2025.
Results of Operations for the Three Months Ended June 30, 2025 and 2024
The following table sets forth our results of operations for the three months ended June 30, 2025 and 2024.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
June 30, |
|
|
Period-to-Period |
|
|
|
2025 |
|
|
2024 |
|
|
Change |
|
|
|
(in thousands) |
|
License and other revenue |
|
$ |
492 |
|
|
$ |
90 |
|
|
$ |
402 |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
Research and development |
|
|
1,584 |
|
|
|
4,603 |
|
|
|
(3,019 |
) |
General and administrative |
|
|
2,476 |
|
|
|
3,077 |
|
|
|
(601 |
) |
Total operating expenses |
|
|
4,060 |
|
|
|
7,680 |
|
|
|
(3,620 |
) |
Loss from operations |
|
|
(3,568 |
) |
|
|
(7,590 |
) |
|
|
4,022 |
|
Interest income |
|
|
115 |
|
|
|
419 |
|
|
|
(304 |
) |
Other income, net |
|
|
1,720 |
|
|
|
1,917 |
|
|
|
(197 |
) |
Non-cash interest expense on liability
related to the sales of future royalties |
|
|
(2,762 |
) |
|
|
(2,340 |
) |
|
|
(422 |
) |
Net loss |
|
|
(4,495 |
) |
|
|
(7,594 |
) |
|
|
3,099 |
|
License and other revenue. In the three months ended June 30, 2025 and 2024, we recognized $0.5 million and $90,000, respectively, of revenue associated with our license agreements. License revenue and other revenue in both periods consisted of revenue for training, services and the sales of our SCS Microinjector kits to our licensees.
Research and development. Research and development expenses decreased by $3.0 million from $4.6 million for the three months ended June 30, 2024 to $1.6 million for the three months ended June 30, 2025. This decrease was primarily due to a $1.6 million decrease in costs related to the CLS-AX program due to the completion of ODYSSEY, our Phase 2b clinical trial, a $0.8 million decrease in employee related costs and $0.4 million decrease related to a research and development tax credit received in the three months ended June 20, 2025.
General and administrative. General and administrative expenses decreased by $0.6 million from $3.1 million for the three months ended June 30, 2024 to $2.5 million for the three months ended June 30, 2025. This decrease was primarily due to a $0.6 million decrease in employee related costs.
Interest income. Interest income decreased by $0.3 million from $0.4 million for the three months ended June 30, 2024 to $0.1 million for the three months ended June 30, 2025. The decrease was due to the lower balance of our cash and cash equivalents.
Other income, net. Other income, net was $1.7 million for the three months ended June 30, 2025 and $1.9 million for the three months ended June 30, 2024. The change was due to a smaller decline in the fair value of the warrant liabilities from the December 31, 2024 valuation date to June 30, 2025 than as compared to the decline in fair value from the February 2024 issuance date to June 30, 2024.
Non-cash interest expense on liability related to the sales of future royalties. Non-cash interest expense on liability related to the sales of future royalties was $2.8 million and $2.3 million for the three months ended June 30, 2025 and 2024, respectively, and was comprised of imputed interest on the liability related to the sales of future royalties and the amortization of the associated issuance costs.
Results of Operations for the Six Months Ended June 30, 2025 and 2024
The following table sets forth our results of operations for the six months ended June 30, 2025 and 2024.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended
June 30, |
|
|
Period-to-Period |
|
|
|
2025 |
|
|
2024 |
|
|
Change |
|
|
|
(in thousands) |
|
License and other revenue |
|
$ |
2,822 |
|
|
$ |
320 |
|
|
$ |
2,502 |
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
Cost of goods sold |
|
|
248 |
|
|
|
— |
|
|
|
248 |
|
Research and development |
|
|
6,047 |
|
|
|
10,218 |
|
|
|
(4,171 |
) |
General and administrative |
|
|
5,300 |
|
|
|
5,901 |
|
|
|
(601 |
) |
Total operating expenses |
|
|
11,595 |
|
|
|
16,119 |
|
|
|
(4,524 |
) |
Loss from operations |
|
|
(8,773 |
) |
|
|
(15,799 |
) |
|
|
7,026 |
|
Interest income |
|
|
278 |
|
|
|
767 |
|
|
|
(489 |
) |
Other income, net |
|
|
1,927 |
|
|
|
418 |
|
|
|
1,509 |
|
Non-cash interest expense on liability
related to the sales of future royalties |
|
|
(5,435 |
) |
|
|
(4,743 |
) |
|
|
(692 |
) |
Loss before income taxes |
|
|
(12,003 |
) |
|
|
(19,357 |
) |
|
|
7,354 |
|
Income tax expense |
|
|
715 |
|
|
|
— |
|
|
|
715 |
|
Net loss |
|
$ |
(12,718 |
) |
|
$ |
(19,357 |
) |
|
$ |
6,639 |
|
License and other revenue. In the six months ended June 30, 2025 and 2024, we recognized $2.8 million and $0.3 million, respectively, of revenue associated with our license agreements. License revenue and other revenue for the six months ended June 30, 2025 consisted of $1.5 million in milestones from Arctic Vision and $1.3 million in other revenue for training, services and the sales of our SCS Microinjector kits to our licensees. License revenue and other revenue for the six months ended June 30, 2024 consisted of $0.3 million for training, services and the sales of our SCS Microinjector kits to our licensees.
Cost of goods sold. In the six months ended June 30, 2025, we recognized $0.2 million in cost of goods sold related to the sales of our SCS Microinjector kits to our licensees of approved products.
Research and development. Research and development expenses decreased by $4.2 million from $10.2 million for the six months ended June 30, 2024 to $6.0 million for the six months ended June 30, 2025. This decrease was primarily due to a $3.4 million decrease in costs related to the CLS-AX program due to the completion of ODYSSEY, our Phase 2b clinical trial, a $0.5 million decrease in employee related costs, a 0.4 million decrease in consulting fees and $0.4 million decrease related to a research and development tax credit received in the six months ended June 30, 2025. This decrease was partially offset by a $0.4 million increase in costs related to the GA program.
General and administrative. General and administrative expenses decreased by $0.6 million from 5.9 million for the six months ended June 30, 2024 to $5.3 million for the six months ended June 30, 2025. This decrease was primarily due to a $0.6 million decrease in employee related costs.
Interest income. Interest income decreased by $0.5 million from $0.8 million for the six months ended June 30, 2024 to $0.3 million for the six months ended June 30, 2025. The decrease was due to the lower balance of our cash and cash equivalents.
Other income, net. Other income, net was $1.9 million for the six months ended June 30, 2025 and $0.4 million for the six months ended June 30, 2024. The change was due to a decline in in the fair value of the warrant liabilities from the December 31, 2024 valuation date to June 30, 2025 than as compared to the decline in fair value from the February 2024 issuance date to June 30, 2024.
Non-cash interest expense on liability related to the sales of future royalties. Non-cash interest expense on liability related to the sales of future royalties was $5.4 million and $4.7 million for the six months ended June 30, 2025 and 2024, respectively, and was comprised of imputed interest on the liability related to the sales of future royalties and the amortization of the associated issuance costs.
Income tax expense. Income tax expense is due to taxes on milestone revenue incurred in foreign jurisdictions.
Liquidity and Capital Resources
Sources of Liquidity
We have funded our operations primarily through the proceeds of public offerings of our common stock, sales of convertible preferred stock, issuance of warrants and the issuance of long-term debt. As of June 30, 2025, we had cash and cash equivalents of
$9.4 million. We invest any cash in excess of our immediate requirements primarily with a view to liquidity and capital preservation. As of June 30, 2025, our funds were held in cash and money market funds.
On February 6, 2024, we entered into a securities purchase agreement, pursuant to which we issued and sold, in a registered direct offering: (i) an aggregate of 11,111,111 shares of our common stock; and (ii) warrants to purchase up to 11,111,111 shares of common stock, or Warrants. The combined purchase price of each share and accompanying Warrant was $1.35. The exercise price for the Warrants is $1.62 per share. The Warrants are currently exercisable and will expire on August 9, 2029. The net proceeds to us from the Registered Direct Offering were $13.9 million.
On January 31, 2024, or the Amendment Effective Date, we entered into a fourth amendment to the license agreement, or the Emory License Agreement, with Emory University and Georgia Tech Research Corporation, or, collectively, the Licensor, pursuant to which the parties agreed to reduce the Sublicense Percentage (as defined in the Emory License Agreement) from a low double digit percentage to a high single digit percentage that we will pay the Licensor applicable to any fees or payments paid to us by any Sublicensee (as defined in the Emory License Agreement) of the Licensed Patents and/or Licensed Technology (each as defined in the Emory License Agreement), excluding (i) amounts paid to us by a Sublicensee to reimburse us for certain research and development costs pursuant to a written agreement between us and such Sublicensee, (ii) the value of intellectual property transferred or granted to us if necessary or helpful to the development or commercialization of Licensed Products (as defined in the Emory License Agreement) and (iii) amounts paid for shares of our stock. The payment to Licensor of any such Sublicense Percentage is due within 30 days of receipt by us of a qualifying payment from a Sublicensee, provided however, with respect to any qualifying payments received by us from a Sublicensee prior to January 1, 2025, the payment to Licensor of any such Sublicensee Percentage is due to Licensor by March 31, 2025. The parties also agreed to a revised annual license maintenance fee due each year, or the Maintenance Fee, starting in 2023 through 2028, as follows: $0.3 million for 2023 through 2025, $0.4 million for each of 2026 and 2027 and $0.5 million for 2028. We paid the Maintenance Fee for 2023 in February 2024 and the Maintenance Fee for 2024 in October 2024. The remaining annual Maintenance Fee payments are due on October 1st of each year.
In May 2023, we entered into a Controlled Equity OfferingSM Sales Agreement, or the Sales Agreement, with Cantor Fitzgerald & Co., or Cantor, under which we may offer and sell, from time to time at our sole discretion, shares of our common stock, having an aggregate offering price of up to $50.0 million through Cantor as our sales agent. During the six months ended June 30, 2025, we sold 1,649,796 shares of our common stock for net proceeds of $1.4 million under the Sales Agreement. During the six months ended June 30, 2024, we sold 339,912 shares of our common stock for net proceeds of $0.5 million under the Sales Agreement.
Funding Requirements
Our primary uses of capital are, and we expect will continue to be, compensation and related expenses, research and development costs to build our product candidate pipeline, legal and other regulatory expenses and general overhead costs as we explore strategic alternatives. In addition, we have certain contractual obligations for future payments. Refer to Note 5 to our consolidated financial statements included in this Quarterly Report on Form 10-Q.
The successful development of our product candidates is highly uncertain. As such, at this time, we cannot reasonably estimate or know the nature, timing and estimated costs of the efforts that will be necessary to complete the remainder of the development of CLS-AX or any future product candidates. We are also unable to predict when, if ever, material net cash inflows will commence from product sales. This is due to the numerous risks and uncertainties associated with developing drugs, including the uncertainty of:
•successful enrollment in, and completion of, clinical trials;
•receipt of marketing approvals from applicable regulatory authorities;
•establishing commercial manufacturing capabilities or making arrangements with third-party manufacturers;
•obtaining and maintaining patent and trade secret protection and regulatory exclusivity for our product candidates; and
•launching commercial sales of the products, if and when approved, whether alone or in collaboration with others.
A change in the outcome of any of these variables with respect to the development of any of our product candidates would significantly change the costs and timing associated with the development of that candidate.
Until such time, if ever, as we can generate substantial product revenue, we may seek to finance our cash needs through a combination of equity offerings, debt financings and potential collaboration, license and development agreements. Other than potential payments we may receive under our license and other agreements, we do not currently have any committed external source of funds, though, as described above, we may also be able to sell our common stock under the Sales Agreement subject to the terms of that agreement and depending on market conditions. We expect that we will require additional capital to fund our ongoing operations. Additional funds may not be available to us on a timely basis, on commercially reasonable terms, or at all. Our ability to raise additional capital may be adversely impacted by potential worsening global economic conditions and disruptions to, and volatility in, the credit and financial markets in the United States and worldwide and related macroeconomic changes, such as rising inflation. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be
diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
If we raise funds through additional collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, including any future collaboration or licensing arrangement for XIPERE outside of the territories in which we have previously licensed or granted options to license XIPERE, we may be required to relinquish additional rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our drug development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
We also incur costs as a public company, including costs and expenses for fees to members of our board of directors, accounting and finance personnel costs, directors and officers insurance premiums, audit and legal fees, investor relations fees and expenses for compliance with reporting requirements under the Exchange Act and rules implemented by the SEC and Nasdaq.
Outlook
We have suffered recurring losses and negative cash flows from operations since inception and anticipate incurring additional losses until such time, if ever, that we can generate significant milestone payments and royalties from XIPERE and other licensing arrangements or revenues from other product candidates. We will need additional financing to fund our operations. Our plans primarily consist of raising additional capital, potentially in a combination of equity or debt financings, monetizing royalties, or restructurings, or potentially entering into additional collaborations, partnerships and other strategic arrangements.
Based on our current plans and forecasted expenses, including the impact of our recent announcement to explore strategic alternatives, we do not believe our cash and cash equivalents will be able to fund our operations for the next 12 months from the date of this filing. In order to conserve our cash balance we have paused all internal research and development programs which can be resumed if we are able to raise additional capital.
These factors raise substantial doubt regarding our ability to continue as a going concern. Our consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might result should we be unable to continue as a going concern.
Cash Flows
The following is a summary of the net cash flows (used in) provided by our operating, investing and financing activities (in thousands):
|
|
|
|
|
|
|
|
|
|
|
Six Months Ended
June 30, |
|
|
|
2025 |
|
|
2024 |
|
Net cash (used in) provided by: |
|
|
|
|
|
|
Operating activities |
|
$ |
(10,559 |
) |
|
$ |
(13,531 |
) |
Investing activities |
|
|
(9 |
) |
|
|
(11,494 |
) |
Financing activities |
|
|
(76 |
) |
|
|
14,343 |
|
Net change in cash and cash equivalents |
|
$ |
(10,644 |
) |
|
$ |
(10,682 |
) |
During the six months ended June 30, 2025 and 2024, our operating activities used net cash of $10.6 million and $13.5 million, respectively. The net cash used in operating activities for the six months ended June 30, 2025 was due to ongoing research and development expenses to develop our pipeline, as well as the supporting general and administrative costs. The net cash used in operating activities for the six months ended June 30, 2024 was due to ongoing research and development expenses to develop our pipeline and costs for ODYSSEY, as well as the supporting general and administrative costs.
During the six months ended June 30, 2025 and 2024, our investing activities used net cash of $9,000 and $11.5 million, respectively. The net cash used in investing activities for the six months ended June 30, 2025 consisted of the acquisition of property and equipment. The net cash used in investing activities for the six months ended June 30, 2024 consisted of $11.0 million for the purchase of short-term investments and $0.5 million for the acquisition of property and equipment.
During the six months ended June 30, 2025 our financing activities used net cash of $76,000. During the six months ended June 30, 2024, our financing activities provided net cash of $14.3 million. The net cash used in financing activities for the six months ended June 30, 2025 consisted primarily of $1.5 million payment related to the royalty purchase and sale agreement, partially offset by proceeds of $1.4 million from the sale of shares of our common stock under the Sales Agreement. The net cash provided by financing activities for the six months ended June 30, 2024 consisted primarily of $13.9 million of net proceeds from the sale of shares of our
common stock and warrants in a registered direct offering and $0.5 million of net proceeds from the sale of shares of our common stock under the Sales Agreement.
Item 3. Quantitative and Qualitative Disclosures about Market Risk
We are a smaller reporting company as defined by Rule 12b-2 of the Exchange Act and are not required to provide the information required under this item.
Item 4. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
The term “disclosure controls and procedures,” as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), refers to controls and procedures that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that such information is accumulated and communicated to a company’s management, including its principal executive and principal financial officers, as appropriate to allow timely decisions regarding required disclosure.
In designing and evaluating our disclosure controls and procedures, management recognizes that disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the disclosure controls and procedures are met. Additionally, in designing disclosure controls and procedures, our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible disclosure controls and procedures. The design of any system of controls also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions; over time, controls may become inadequate because of changes in conditions, or the degree of compliance with policies or procedures may deteriorate. Because of the inherent limitations in a control system, misstatements due to error or fraud may occur and not be detected.
Our management, with the participation of our Chief Executive Officer and our Chief Financial Officer, has evaluated the effectiveness of our disclosure controls and procedures as of the end of the period covered by this report. Based on that evaluation, our Chief Executive Officer and our Chief Financial Officer concluded that our disclosure controls and procedures were effective as of the end of the period covered by this report at the reasonable assurance level.
Changes in Internal Control over Financial Reporting
There has been no change in our internal control over financial reporting during the quarter ended June 30, 2025 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting. We have not experienced any material impact to our internal control over financial reporting.
PART II – OTHER INFORMATION
Item 1. Legal Proceedings
From time to time, we may be subject to litigation and claims arising in the ordinary course of business. We are not currently a party to any material legal proceedings and we are not aware of any pending or threatened legal proceeding against us that we believe could have a material adverse effect on our business, operating results, cash flows or financial condition.
Item 1A. Risk Factors
Our business is subject to risks and events that, if they occur, could adversely affect our financial condition and results of operations and the trading price of our securities. In addition to the other information set forth in this quarterly report on Form 10-Q, you should carefully consider the factors described in “Part I, Item 1A. Risk Factors” of our Annual Report on Form 10-K for the fiscal year ended December 31, 2024, filed with the Securities and Exchange Commission on March 27, 2025.
Risks Related to Our Financial Position and Capital Needs
We do not currently have sufficient working capital to fund our planned operations for the next twelve months and substantial doubt exists as to our ability to continue as a going concern.
Our historical financial statements have been prepared under the assumption that we will continue as a going concern. As of June 30, 2025, we had an accumulated deficit of $368.0 million and had cash and cash equivalents of $9.4 million. While we have begun to implement cost reductions in 2025, we have finite cash resources available to fund our operations. As part of cost reductions, we implemented a plan pursuant to which we terminated all employees and transitioned such persons into consulting roles. We estimate that we will incur charges of approximately $2.6 million for severance and other employee termination-related costs in the third quarter of 2025. The estimated charges that we expect to incur are subject to a number of assumptions, and actual results may differ materially from these estimates. We may also incur additional costs not currently contemplated due to events that may occur as a result of, or that are associated with, our workforce reduction. The reduction in force is expected to be completed during the third quarter of 2025.
In July 2025, we announced plans to explore a full range of strategic alternatives to advance our SCS platform and drug development pipeline to maximize stockholder value. We have retained Piper Sandler, a leading investment bank with substantial experience in the biotechnology industry, to support us with the strategic evaluation process. Strategic alternatives under consideration include the sale, license, monetization and/or divestiture of one or more of our assets and technologies, collaboration, partnership, merger, acquisition, joint ventures or other strategic transactions.
These conditions raise substantial doubt about our ability to continue as a going concern within one year after the date the financial statements are issued. Based on our current plans and forecasted expenses and our cash and cash equivalents, we do not believe we will be able to fund our operations for the next 12 months from the date of this filing. Until we can generate sufficient revenue, if ever, to fund our operations, we will need to finance future cash needs through public or private equity offerings, license agreements, debt financings or restructurings, collaborations, strategic alliances and marketing or distribution arrangements.
The perception of our ability to continue as a going concern may make it more difficult for us to obtain financing for the continuation of our operations and could result in the loss of confidence by investors and employees. Additional financing may not be available to us when needed or, if available, it may not be obtained on commercially reasonable terms. If we are not able to obtain the necessary additional financing and if a strategic alternative is not available, we may be forced to file for bankruptcy or wind down our operations.
Our activities to evaluate and pursue potential strategic alternatives may not result in any transaction or enhance stockholder value.
We have begun evaluating and exploring a variety of strategic alternatives focused on maximizing stockholder value, including, but not limited to, an acquisition, merger, reverse merger, other business combination, sales of assets or other strategic transactions. Our ability to successfully execute on a strategic alternative is dependent on a number of factors and we may not be able to execute upon a transaction or other strategic alternative upon favorable terms within an advantageous timeframe and recognize significant value for our assets, if at all. Additionally, the negotiation and consummation of a transaction or other strategic alternative may be costly and time-consuming. Any executed strategic alternative may not maximize or even enhance stockholder value, could result in total costs and expenses that are greater than expected, could make it more difficult to attract and retain qualified personnel and may disrupt our operations, each of which could have a material adverse effect on our business.
The market price of our common stock may reflect a market assumption that a strategic alternative will occur, and a failure to complete a strategic alternative could result in negative investor perceptions and could cause a decline in the market price of our common stock, which could adversely affect our ability to access the equity and financial markets, as well as our ability to explore and
enter into different strategic alternatives. There can be no certainty that any strategic alternative will be completed, be on attractive terms, enhance stockholder value or deliver the anticipated benefits, and successful integration or execution of the strategic alternatives will be subject to additional risks. In addition, potential strategic alternatives that require stockholder approval may not be approved by our stockholders. If we do not successfully consummate a strategic alternative, our board of directors may decide to pursue a dissolution and liquidation of our company. In such an event, the amount of cash available for distribution to our stockholders will depend heavily on the timing of such liquidation, the amount of cash that will need to be reserved for commitments and contingent liabilities. Depending on these factors, the amount available for distribution to our common stockholders could be as low as zero and result in a total loss of investment to our stockholders.
Risks Related to Ownership of Our Common Stock
If we fail to meet all applicable requirements of Nasdaq and Nasdaq determines to delist our common stock, the delisting could adversely affect the market liquidity of our common stock and the market price of our common stock could decrease.
On February 7, 2025, we received a letter from Nasdaq, notifying us that, from December 18, 2024 to February 4, 2025, the closing bid price for our common stock was below $1.00. In accordance with Nasdaq Listing Rule 5810(c)(3)(A) we were provided an initial period of 180 calendar days, or until August 6, 2025, to regain compliance with Nasdaq’s Minimum Bid Price Requirement. We did not regain compliance by August 6, 2025, and as a result we expect that Nasdaq will provide notice that our common stock will be subject to delisting. We intend to appeal any delisting determination to a Nasdaq hearings panel and our common stock will remain listed on the Nasdaq Global Market until the completion of the appeal process, during which time we may regain compliance the Minimum Bid Price Requirement if our minimum bid price of listed securities closes at $1.00 per share or more for a minimum of 10 consecutive business days. In addition, on August 4, 2025, we filed a proxy statement seeking approval by our stockholders to effect a reverse split of our outstanding common stock at a ratio ranging from 1-for-5 and 1-for-15. If approved by stockholders, this Reverse Stock Split Proposal would permit (but not require) our board of directors to effect a reverse stock split of our common stock at any time before August 29, 2026, by a ratio of not less than 1-for-5 and not more than 1-for-15, inclusive, and a corresponding proportionate reduction in the total number of authorized shares of the Company’s common stock, with such ratio to be determined by the our board of directors in its sole discretion. Our board of directors’ primary objective in proposing the Reverse Stock Split and Authorized Shares Reduction is to raise the per share trading price of our common stock. Our board of directors believes that the Reverse Stock Split and Authorized Shares Reduction will result in a higher per share trading price, which is intended to enable us to maintain the listing of our common stock on The Nasdaq Global Market and generate greater investor interest in the Company.
There can be no assurance that we will regain compliance with the requirements for listing our common stock on Nasdaq. If we are unable to satisfy the Nasdaq criteria for continued listing, our common stock would be subject to delisting. A delisting of our common stock could negatively impact us by, among other things, reducing the liquidity and market price of our common stock; reducing the number of investors willing to hold or acquire our common stock, which could negatively impact our ability to raise equity financing; decreasing the amount of news and analyst coverage of us; and limiting our ability to issue additional securities or obtain additional financing in the future. In addition, delisting from Nasdaq may negatively impact our reputation and, consequently, our business.
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds
Sales of Unregistered Securities
None.
Issuer Purchases of Equity Securities
None.
Item 5. Other Information
During the quarter ended June 30, 2025, none of our directors or officers (as defined in Rule 16a-1(f) under the Exchange Act) adopted, modified or terminated a “Rule 10b5-1 trading arrangement” or a “non-Rule 10b5-1 trading arrangement” (as each term is defined in Item 408 of Regulation S-K).
Item 6. Exhibits
|
|
|
Exhibit No. |
|
Description |
|
|
|
3.1 |
|
Amended and Restated Certificate of Incorporation (incorporated herein by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K (File No. 001-37783) filed with the SEC on June 7, 2016). |
|
|
|
3.2 |
|
Certificate of Amendment to Amended and Restated Certificate of Incorporation (incorporated herein by reference to Exhibit 3.1 to the Registrant's Current Report on Form 8-K (File No. 001-37783) filed with the SEC on June 23, 2022). |
|
|
|
3.3 |
|
Certificate of Amendment to Amended and Restated Certificate of Incorporation (incorporated herein by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K (File No. 001-37783) filed with the SEC on May 30, 2025). |
|
|
|
3.4 |
|
Amended and Restated Bylaws (incorporated herein by reference to Exhibit 3.2 to the Registrant's Current Report on Form 8-K (File No. 001-37783) filed with the SEC on June 7, 2016). |
|
|
|
10.1* |
|
Separation Agreement, by and between Registrant and George Lasezkay, dated as of July 17, 2025. |
|
|
|
10.2* |
|
Consulting Agreement, by and between Registrant and George Lasezkay, dated as of July 17, 2025. |
|
|
|
10.3* |
|
Separation Agreement, by and between Registrant and Charles Deignan, dated as of July 17, 2025. |
|
|
|
10.4* |
|
Consulting Agreement, by and between Registrant and Charles Deignan, dated as of July 17, 2025. |
|
|
|
10.5* |
|
Separation Agreement, by and between Registrant and Victor Chong, dated as of July 17, 2025. |
|
|
|
10.6* |
|
Consulting Agreement, by and between Registrant and Victor Chong, dated as of July 17, 2025. |
|
|
|
31.1* |
|
Certification of Principal Executive Officer under Section 302 of the Sarbanes-Oxley Act. |
|
|
|
31.2* |
|
Certification of Principal Financial Officer under Section 302 of the Sarbanes-Oxley Act. |
|
|
|
32.1** |
|
Certifications of Principal Executive Officer and Principal Financial Officer under Section 906 of the Sarbanes-Oxley Act. |
|
|
|
101.INS |
|
Inline XBRL Instance Document |
|
|
|
101.SCH |
|
Inline XBRL Taxonomy Extension Schema Document with Embedded Linkbase Documents |
|
|
|
104 |
|
Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibits 101) |
|
|
* |
Filed herewith. |
** |
These certifications are being furnished solely to accompany this quarterly report pursuant to 18 U.S.C. Section 1350, and are not being filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, and are not to be incorporated by reference into any filing of the registrant, whether made before or after the date hereof, regardless of any general incorporation language in such filing. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
|
|
Clearside Biomedical, Inc. |
|
|
|
|
Date: August 8, 2025 |
|
By: |
/s/ Charles A. Deignan |
|
|
|
Charles A. Deignan |
|
|
|
Chief Financial Officer |
|
|
|
(On behalf of the Registrant and as
Principal Financial Officer) |
EX-10.1

July 17, 2025
Via Email
George Lasezkay
Re: Separation Agreement
Dear George:
This letter sets forth the substance of the separation agreement (the “Agreement”) which Clearside Biomedical, Inc. (the “Company”) is offering to you to aid in your employment transition.
1.Separation Date. Your last day of work with the Company and your employment termination date will be July 18, 2025 (the “Separation Date”). Between the date of this Agreement and your Separation Date, you will be expected to perform your regular job duties and such other duties as may be assigned to you by the Company’s Board of Directors (the “Board”). Notwithstanding anything to the contrary in Section 5(e) of your Employment Agreement (as defined below), you may continue your service as a director on the Board following your Separation Date.
2.Accrued Salary. In accordance with applicable law, the Company will pay you all accrued salary earned through the Separation Date subject to standard payroll deductions and withholdings. You acknowledge that you will receive these payments regardless of whether or not you execute this Agreement.
3.Severance Benefits. In lieu of the severance benefits you are eligible to receive pursuant to Section 5 of your March 11, 2020 Amended and Restated Executive Employment Agreement between you and the Company (the “Employment Agreement”), and provided you return this fully signed and dated Agreement to the Company within the time frame specified below (but no earlier than the Separation Date) do not revoke it, and fully comply with your obligations under this Agreement, and under your previously executed Confidential Information Agreement (as defined below) (collectively, the “Severance Preconditions”), the Company will, provide you with the following severance benefits (the “Severance Benefits”):
(a) Severance. The Company will pay you, as severance, the equivalent of thirty-nine (39) weeks of your base salary in effect as of the Separation Date, subject to standard payroll deductions and withholdings. This amount will be paid in a lump sum on the Company’s next regular payroll date following the Effective Date (as defined below), provided you have signed and not revoked the Agreement by such date.
(b) COBRA Severance Benefit. If you timely elect continued coverage under COBRA under the Company’s group health plans, then, as an additional severance benefit, the Company will cover the cost of your full COBRA premiums to continue your coverage (including coverage for eligible dependents, if applicable) in effect for yourself (and your eligible dependents, if applicable) until the earliest of: (A) nine (9) months following the Separation Date; (B) the expiration of your eligibility for the continuation coverage under COBRA; or (C) the date when you become eligible for and covered by substantially equivalent health insurance coverage in connection with new employment or self-employment (such period from the termination date through the earliest of (A) through (C), the “COBRA Payment Period” and such severance benefit, the “COBRA Severance Benefit”). Notwithstanding the foregoing, if at any time, (i) the Company determines, in its sole discretion, that the payment of the COBRA premiums would result in a violation of the nondiscrimination rules of Section 105(h)(2) of the Internal Revenue Code or any statute or regulation of similar effect (including but not limited to the 2010 Patient Protection and Affordable Care Act, as amended by the 2010 Health Care and Education Reconciliation Act), or (ii) the Company’s health insurance plan in effect on the Separation Date terminates (either (i) or (ii), a “COBRA Severance Benefit Terminating Event”), then provided you otherwise were eligible for the COBRA Severance Benefit on the date of the COBRA Severance Benefit Terminating Event, in lieu of providing the COBRA premium reimbursement, the Company will instead pay you a fully taxable lump sum cash payment equal to the remainder of the COBRA premiums due under this Section 3(b) for the COBRA Payment Period (the “Special Cash Payment”), which payment shall be payable within 30 days following the applicable COBRA Severance Benefit Terminating Event. You may, but are not obligated to, use such Special Cash Payment for medical expenses, including COBRA Premiums (as applicable). If, prior to any COBRA Severance Benefit Terminating Event, you become eligible for coverage under another employer’s group health plan through self-employment or otherwise cease to be eligible for COBRA coverage under the Company’s group health plan during the COBRA Payment Period, you must immediately notify the Company of such event, and all payments and obligations under this Section will cease.
(c) Consulting Agreement. The Company will continue to retain you in a consulting role pursuant to the Consulting Agreement attached as Exhibit A (the “Consulting Agreement”), which shall be deemed effective as of the Separation Date. Notwithstanding the foregoing, if you do not timely sign and return this Agreement, or you sign but later revoke your acceptance of this Agreement, the Consulting Agreement will terminate in accordance with its terms.
(d) PTEP Extension. The Company will, subject to the approval of the Board, extend the period of time during which you may exercise any vested, outstanding and unexercised stock Options (as defined below) until the earlier of: (i) the date that is twelve (12) months following the termination of your Continuous Service (as that term is defined in the Plan (as defined below)), or (ii) the original expiration date applicable to each of the Options, unless terminated earlier in accordance with the terms of the Plan and the Equity Award Documents. Except as provided in this Agreement, all terms, conditions and limitations applicable to the Options will remain in full force and effect pursuant to the Plan and the Equity Award Documents. The Company makes no representations or guarantees regarding the status of your Options as incentive stock options (“ISOs”). You understand and agree if any Option that otherwise qualifies as an ISO is exercised with respect to any vested shares later than the date that is three (3) months following the Separation Date, such Option will be treated as a non-qualified stock option (“NSO”), and you will
be obligated to satisfy your tax obligations that arise when you exercise such Option. No shares of the Company’s common stock will be issued to you in respect of any Options treated as NSOs unless and until you satisfy such tax obligations. You acknowledge that the Company is not providing tax advice to you and that you have been advised by the Company to seek independent tax advice with respect to the exercise and modification of the Options.
(e) Retention of Laptop Computer. As an additional Severance Benefit, the Company will permit you to retain the Company-issued laptop computer provided to you during your employment; provided, however, that you agree to reasonably cooperate with the Company to confirm all files stored on the computer are intact and all Company confidential information and trade secrets have been deleted after the end of the Consulting Period (as defined in the Consulting Agreement).
(f) Letter of Reference. As an additional Severance Benefit, upon your request, the Company will provide you with a signed letter of reference. This letter of reference will be provided to you after you send a written request for the letter to the Company, provided you have returned this executed Agreement to the Company by the date of such request.
The Company is offering severance to you in reliance on Treasury Regulation Section 1.409A-1(b)(9) and the short-term deferral exception in Treasury Regulation Section 1.409A-1(b)(4). Any payments made in reliance on Treasury Regulation Section 1.409A-1(b)(4) will be made not later than March 15 of the year following the year in which the Separation Date occurs. For purposes of Section 409A of the Internal Revenue Code of 1986, as amended (“Code Section 409A”), your right to receive any installment payments under this letter (whether severance payments, reimbursements or otherwise) shall be treated as a right to receive a series of separate payments and, accordingly, each installment payment hereunder shall at all times be considered a separate and distinct payment. To the extent that any Severance Benefits are not otherwise exempt from the application of Code Section 409A, then, if the period during which you may consider and sign this Agreement spans two calendar years, the payment of severance will not be made or begin until the later calendar year.
4.Benefit Plans. If you are currently participating in the Company’s group health insurance plans, including medical, dental, and/or vision plans, your participation as an employee will end on last day of the month in which the Separation Date occurred. Thereafter, to the extent provided by the federal COBRA law or, if applicable, state insurance laws, and by the Company’s current group health insurance policies, you will be eligible to continue your group health insurance benefits at your own expense, with the potential for certain payments to be made by the Company as described in Section 3(b) above. For questions on your benefits, you may reach out to TriNet Solution Center at 800-638-0461 or employees@TriNet.com.
5.Unemployment Insurance. You may be eligible for unemployment insurance benefits after the Separation Date. You acknowledge that whether you receive unemployment compensation will be decided by the applicable agency that is charged with unemployment insurance matters in your state, and not by the Company. That agency can provide you with benefits and eligibility information regarding unemployment compensation.
6.Equity Awards. If you were granted stock options to purchase certain shares of the Company’s common stock (the “Options”) and/or restricted stock units to be issued shares of the Company’s common stock (the “RSUs” and together with the Options, the “Equity Awards”) pursuant to the Company’s 2016 Equity Incentive Plan (the “Plan”) and your applicable Option and/or RSU agreement (together with the Plan, the “Equity Award Documents”), vesting of your Options and/or RSUs (as applicable) will cease as of the date your “Continuous Service” (as defined in the Plan) ends. For avoidance of doubt, if you satisfy the Severance Preconditions, then the Equity Awards remain and will continue to remain outstanding and the unvested shares subject to the Equity Awards will continue to be eligible to vest following the Separation Date during the Consulting Period (as defined in the Consulting Agreement), in accordance with the vesting schedules applicable to such Equity Awards and subject to your Continuous Service during such periods. Any Options or RSUs that you currently hold shall continue to be governed by the terms of the Equity Award Documents.
7.Other Compensation or Benefits. You acknowledge that, except as expressly provided in this Agreement and the Consulting Agreement, you will not receive any additional compensation, severance, commissions, bonuses or benefits after the Separation Date. You also acknowledge that the Company’s provision of the Severance Benefits set forth in Section 3 above fully satisfy any severance or other obligations under the Employment Agreement.
8.Expense Reimbursements. Within ten (10) days following the Separation Date, you agree to submit your final documented expense reimbursement statement reflecting all business expenses you incurred through the Separation Date, if any, for which you seek reimbursement. The Company will reimburse you for reasonable business expenses pursuant to its regular business practice.
9.Return of Company Property. Within three (3) days following the end of the Consulting Period, you agree to return to the Company all Company documents (and all copies thereof) and other Company property that you have had in your possession at any time, including, but not limited to, Company files, notes, drawings, records, business plans and forecasts, financial information, specifications, computer-recorded information, tangible property (other than the laptop referenced in Section 3 above), credit cards, entry cards, identification badges and keys; and, any materials of any kind that contain or embody any proprietary or confidential information of the Company (and all reproductions thereof).
10.Proprietary Information and Post-Termination Obligations. Both during and after your employment you acknowledge your continuing obligations under your Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement or any other confidentiality agreement you signed with the Company (the “Confidential Information Agreement”) not to use or disclose any confidential or proprietary information of the Company and to refrain from certain other activities. A copy of the Confidential Information Agreement is attached hereto as Exhibit B. As you know, the Company will enforce its contract rights. Please familiarize yourself with the enclosed agreement which you signed. Confidential information that is also a “trade secret,” as defined by law, may be disclosed (A) if it is made (i) in confidence to a federal, state, or local government official, either directly or indirectly, or to an attorney and (ii) solely for the purpose of reporting or investigating a suspected violation of law; or (B) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under
seal. In addition, in the event that you file a lawsuit for retaliation by the Company for reporting a suspected violation of law, you may disclose the trade secret to your attorney and use the trade secret information in the court proceeding, if you: (A) file any document containing the trade secret under seal; and (B) do not disclose the trade secret, except pursuant to court order.
11. Mutual Non-Disparagement. Both you and the Company agree not to disparage the other party, and the other party’s officers, directors, employees, shareholders and agents, in any manner reasonably likely to be harmful to them or their business, business reputation or personal reputation. The Company’s obligation not to disparage you under this Section are limited to the Company’s current officers and directors. Notwithstanding the foregoing, nothing in this Agreement shall limit your right to voluntarily communicate with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, other federal government agency or similar state or local agency or to discuss the terms and conditions of your employment with others to the extent expressly permitted by Section 7 of the National Labor Relations Act. In addition, nothing in this Section or this Agreement is intended to prohibit or restrain you in any manner from making disclosures protected under the whistleblower provisions of federal or state law or regulation or other applicable law or regulation.
12.Cooperation after Termination. Consistent with your obligations under the Employment Agreement, you agree to cooperate fully with the Company in all matters relating to the transition of your work and responsibilities on behalf of the Company, including, but not limited to, any present, prior or subsequent relationships and the orderly transfer of any such work and institutional knowledge to such other persons as may be designated by the Company, by making yourself reasonably available during regular business hours.
(a)General Release of Claims. In exchange for the consideration provided to you under this Agreement to which you would not otherwise be entitled, you hereby generally and completely release the Company, and its affiliated, related, parent and subsidiary entities, and its and their current and former directors, officers, employees, shareholders, partners, agents, co-employers, including TriNet Group, Inc., attorneys, predecessors, successors, insurers, affiliates, and assigns (the “Company Parties”) from any and all claims, liabilities, demands, causes of action, and obligations, both known and unknown, arising from or in any way related to events, acts, conduct, or omissions occurring at any time prior to and including the date you sign this Agreement.
(b)Scope of Release. This general release includes, but is not limited to: (i) all claims arising from or in any way related to your employment with the Company or the termination of that employment; (ii) all claims related to your compensation or benefits from the Company, including salary, bonuses, commissions, vacation pay, expense reimbursements, severance pay, fringe benefits, stock, stock options, or any other ownership, equity, or profits interests in the Company; (iii) all claims for breach of contract, wrongful termination, and breach of the implied covenant of good faith and fair dealing; (iv) all tort claims, including claims for fraud, defamation, emotional distress, and discharge in violation of public policy; and (v) all federal, state, and local statutory claims, including claims for discrimination, harassment, retaliation, attorneys’ fees, or
other claims arising under the federal Civil Rights Act of 1964 (as amended), the federal Americans with Disabilities Act of 1990, the Age Discrimination in Employment Act, as amended (“ADEA”), the Georgia Fair Employment Practices Act, the Georgia Equal Pay Act, the Georgia Age Discrimination in Employment Law, the Georgia Equal Employment for Persons with Disabilities Code, the Georgia Minimum Wage Law, the Georgia Equal Pay Act, the Georgia Law on Discrimination on the Basis of Maternity Leave, and the Georgia Law on Sex Discrimination.
(c)ADEA Release. You acknowledge that you are knowingly and voluntarily waiving and releasing any rights you have under the ADEA, and that the consideration given for the waiver and releases you have given in this Agreement is in addition to anything of value to which you were already entitled. You further acknowledge that you have been advised, as required by the ADEA, that: (i) your waiver and release does not apply to any rights or claims arising after the date you sign this Agreement; (ii) you should consult with an attorney prior to signing this Agreement (although you may choose voluntarily not to do so); (iii) you have forty-five (45) days to consider this Agreement (although you may choose voluntarily to sign it sooner); (iv) you have seven (7) days following the date you sign this Agreement to revoke this Agreement (in a written revocation sent to the Company); and (v) this Agreement will not be effective until the date upon which the revocation period has expired, which will be the eighth day after you sign this Agreement provided that you do not revoke it. You also hereby acknowledge that the Company has provided you with the ADEA Disclosure Schedule (pursuant to Title 29 U.S. Code Section 626(f)(1)(H)), attached as Exhibit C to this Agreement.
(d)Exceptions. Notwithstanding the foregoing, you are not releasing the Company hereby from: (i) any claims that may arise from events that occur after the date this waiver is executed; (ii) any existing obligation to indemnify you pursuant to the Articles and Bylaws of the Company, any valid fully executed indemnification agreement with the Company, applicable law, or applicable directors and officers liability insurance; (iii) any claims that cannot be waived by law, including, without limitation, any rights you may have under applicable workers’ compensation laws and your right, if applicable, to file or participate in an investigative proceeding of any federal, state, or local government agency; or (iv) any claims for breach of this Agreement.
14.Protected Rights. You understand that nothing in this Agreement has limited, currently limits, or shall limit your ability to file a charge or complaint with the Equal Employment Opportunity Commission, the Department of Labor, the National Labor Relations Board, the Occupational Safety and Health Administration, the Department of Justice, the Securities and Exchange Commission, or any other government agency, law enforcement agency, or commission (“Government Agencies”). You further understand this Agreement does not limit your ability to communicate with any Government Agencies or otherwise participate in any investigation or proceeding that may be conducted by any Government Agency, including providing documents or other information, without notice to the Company. While this Agreement does not limit your right to receive a government-issued award for information provided to any Government Agency in connection with a government whistleblower program or protected whistleblower activity, you understand and agree that, to maximum extent permitted by law, you are otherwise waiving any and all rights you may have to individual relief based on any claims that you have released and any rights you have waived by signing this Agreement. Nothing in this Agreement has prevented, currently prevents, or shall prevent you from discussing or disclosing information about unlawful
acts in the workplace, such as harassment, or discrimination or any other conduct that you have reason to believe is unlawful. Additionally, nothing in this Agreement waives any rights you may have under Section 7 of the National Labor Relations Act (subject to the release of claims set forth herein).
15.Your Acknowledgments and Affirmations. You acknowledge and agree that (i) the consideration given to you in exchange for the waiver and release in this Agreement is in addition to anything of value to which you were already entitled; and (ii) that you have been paid for all time worked, have received all the leave, leaves of absence and leave benefits and protections for which you are eligible, and have not suffered any on-the-job injury for which you have not already filed a claim. You affirm that all of the decisions of the Company Parties regarding your pay and benefits through the date of your execution of this Agreement were not discriminatory based on age, disability, race, color, sex, religion, national origin or any other classification protected by law. You affirm that you will not voluntarily (except in response to legal compulsion or as permitted in Section 15 above) assist any person in bringing or pursuing any proposed or pending litigation, arbitration, administrative claim or other formal proceeding against any of the Company Parties. You further affirm that you have no known workplace injuries or occupational diseases. You acknowledge and affirm that you have not been retaliated against for reporting any allegation of corporate fraud or other wrongdoing by any of the Company Parties, or for exercising any rights protected by law, including any rights protected by the Fair Labor Standards Act, the Family Medical Leave Act, or any related statute or local leave or disability accommodation laws, or any applicable state workers’ compensation law.
16.No Admission. This Agreement does not constitute an admission by the Company of any wrongful action or violation of any federal, state, or local statute, or common law rights, including those relating to the provisions of any law or statute concerning employment actions, or of any other possible or claimed violation of law or rights.
17.Breach. You agree that upon any breach of this Agreement you will forfeit all amounts paid or owing to you under this Agreement. Further, you acknowledge that it may be impossible to assess the damages caused by your violation of the terms of Sections 9, 10, 11, and 12 of this Agreement and further agree that any threatened or actual violation or breach of those Sections of this Agreement will constitute immediate and irreparable injury to the Company. You therefore agree that any such breach of this Agreement is a material breach of this Agreement, and, in addition to any and all other damages and remedies available to the Company upon your breach of this Agreement, the Company shall be entitled to an injunction to prevent you from violating or breaching this Agreement. You agree that if the Company is successful in whole or part in any legal or equitable action against you under this Agreement, you agree to pay all of the costs, including reasonable attorneys’ fees, incurred by the Company in enforcing the terms of this Agreement.
18.Dispute Resolution. You and the Company agree that any and all disputes, claims, or controversies of any nature whatsoever arising from, or relating to, this Agreement or its interpretation, enforcement, breach, performance, or execution, or any of the matters herein released (collectively, “Claims,” each a “Claim”), shall be resolved, pursuant to the Federal Arbitration Act, 9 U.S.C. §1-16, and to the fullest extent permitted by law, by final, binding, and confidential arbitration at a location closest to where you last worked for the Company or another
mutually agreeable location. The arbitration shall be conducted before a single neutral arbitrator by JAMS, Inc. (“JAMS”) or its successor, under the then applicable JAMS Comprehensive Arbitration Rules and Procedures (currently available at https://www.jamsadr.com/rules-comprehensive-arbitration) (“JAMS Rules”) and Georgia law. Both you and the Company opt into the Expedited Procedures under the JAMS Rules. The arbitrator shall apply substantive and procedural Georgia law to any dispute or claim, without reference to any conflict-of-law provisions of any jurisdiction. To the extent that the JAMS Rules conflict with Georgia law, Georgia law shall take precedence. The parties agree that punitive damages shall not be available in arbitration. By agreeing to this arbitration procedure, both you and the Company waive the right to have any Claim resolved through a trial by jury or judge or an administrative proceeding. This Section shall not apply to any action or claim that cannot be subject to mandatory arbitration as a matter of law. The arbitrator shall have sole authority for determining if a Claim is subject to arbitration, and any other procedural questions related to the dispute and bearing on the final disposition. In addition, the arbitrator shall: (a) have the authority to compel adequate discovery for the resolution of the dispute and to award such relief as would otherwise be available under applicable law in a court proceeding; and (b) issue a written statement signed by the arbitrator regarding the disposition of each claim and the relief, if any, awarded as to each claim, the reasons for the award, and the arbitrator’s essential findings and conclusions on which the award is based. You and the Company shall each pay half the costs and expenses of the arbitration and each pay for its respective attorneys’ fees and costs, except as prohibited by law. To the extent JAMS does not collect or you otherwise do not pay to JAMS an equal share of all JAMS’ arbitration fees for any reason, and the Company pays JAMS your share, you acknowledge and agree that the Company shall be entitled to recover from you half of the JAMS arbitration fees invoiced to the parties (less any amounts you paid to JAMS) in a federal or state court of competent jurisdiction. Nothing in this letter agreement is intended to prevent either you or the Company from obtaining injunctive relief in court to prevent irreparable harm pending the conclusion of any such arbitration. Any awards or orders in such arbitrations may be entered and enforced as judgments in the federal and state courts of any competent jurisdiction.
_____ By initialing here, YOU acknowledge YOU have read and agree to this arbitration provision.
19.Miscellaneous. This Agreement, including Exhibits A, B and C, constitutes the complete, final and exclusive embodiment of the entire agreement between you and the Company with regard to this subject matter. It is entered into without reliance on any promise or representation, written or oral, other than those expressly contained herein, and it supersedes any other such promises, warranties or representations. This Agreement may not be modified or amended except in a writing signed by both you and a duly authorized officer of the Company. This Agreement will bind the heirs, personal representatives, successors and assigns of both you and the Company, and inure to the benefit of both you and the Company, their heirs, successors and assigns. If any provision of this Agreement is determined to be invalid or unenforceable, in whole or in part, this determination will not affect any other provision of this Agreement and the provision in question will be modified by the court so as to be rendered enforceable. This Agreement will be deemed to have been entered into and will be construed and enforced in accordance with the laws of the state or commonwealth in which you primarily performed work for the Company as applied to contracts made and to be performed entirely within such state or commonwealth.
If this Agreement is acceptable to you, please sign below and return it to me on or before the date that is forty-five (45) days after you receive this Agreement (but no earlier than the Separation Date). The Company’s severance offer contained herein will automatically expire if you do not sign and return the fully signed Agreement within this timeframe.
[SIGNATURES TO FOLLOW ON NEXT PAGE]
Sincerely,
Clearside Biomedical, Inc.
By: /s/Tony Gibney
Tony Gibney
Chair, Board of Directors
I have read, understand and agree fully to the foregoing Agreement. I understand that this Agreement includes a release of all known and unknown claims, even those unknown claims that, if known by me, would affect my decision to accept this Agreement.
/s/George Lasezkay
George Lasezkay
July 19,2025
Date
Exhibit A – Consulting Agreement
Exhibit B – Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C – ADEA disclosure
Exhibit A
Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and George Lasezkay, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services as Client’s Principal Executive Officer, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $350 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief
itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $861,840, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or
any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to
any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade
secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of
Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: ____________________________________
Tony Gibney
Chair, Board of Directors
Agreed to and Accepted:
________________________________________
George Lasezkay
____________________
Date
Exhibit B
Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C
ADEA Disclosure Schedule
The following disclosure was prepared as of July 17, 2025. It shows the employees in the decisional unit whose employment will be terminated pursuant to the business reorganization announced on or around July 17, 2025, and the factors used to determine who was eligible for the layoff. Employees who will be terminated will be eligible for benefits based upon their execution of a waiver. This data is subject to change and may be affected by future employment decisions.
Those receiving this disclosure will have forty-five (45) days to review the terms and conditions of the severance package.
On July 17, 2025, the employment of all individuals employed by the Company will be terminated in the business reorganization and no individuals employed by the Company will be retained.
The factors used to determine eligibility for the layoff are:
The Company’s assessment of its current and projected business and cost-reduction needs to effectuate its plan to cease all its active development activities and assess and execute strategic alternatives to its current business model. Not all criteria were used to make every selection.
EX-10.2

Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and George Lasezkay, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services as Client’s Principal Executive Officer, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $350 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $861,840, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable
time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall
be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such
services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local
agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation
Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to
Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: /s/Tony Gibney
Tony Gibney
Chair, Board of Directors
Agreed to and Accepted:
/s/George Lasezkay
George Lasezkay
July 19, 2025
Date
EX-10.3

July 17, 2025
Via Email
Charlie Deignan
Re: Separation Agreement
Dear Charlie:
This letter sets forth the substance of the separation agreement (the “Agreement”) which Clearside Biomedical, Inc. (the “Company”) is offering to you to aid in your employment transition.
1.Separation Date. Your last day of work with the Company and your employment termination date will be July 18, 2025 (the “Separation Date”). Between the date of this Agreement and your Separation Date, you will be expected to perform your regular job duties and such other duties as may be assigned to you by the Company’s Board of Directors (the “Board”). Notwithstanding anything to the contrary in Section 5(e) of your Employment Agreement (as defined below), you may continue your service as a director on the Board following your Separation Date.
2.Accrued Salary. In accordance with applicable law, the Company will pay you all accrued salary earned through the Separation Date subject to standard payroll deductions and withholdings. You acknowledge that you will receive these payments regardless of whether or not you execute this Agreement.
3.Severance Benefits. In lieu of the severance benefits you are eligible to receive pursuant to Section 5 of your August 3, 2017 Amended and Restated Executive Employment Agreement between you and the Company (the “Employment Agreement”), and provided you return this fully signed and dated Agreement to the Company within the time frame specified below (but no earlier than the Separation Date) do not revoke it, and fully comply with your obligations under this Agreement, and under your previously executed Confidential Information Agreement (as defined below) (collectively, the “Severance Preconditions”), the Company will, provide you with the following severance benefits (the “Severance Benefits”):
(a) Severance. The Company will pay you, as severance, the equivalent of thirty-one (31) weeks of your base salary in effect as of the Separation Date, subject to standard payroll deductions and withholdings. This amount will be paid in a lump sum on the Company’s next
regular payroll date following the Effective Date (as defined below), provided you have signed and not revoked the Agreement by such date.
(b) COBRA Severance Benefit. If you timely elect continued coverage under COBRA under the Company’s group health plans, then, as an additional severance benefit, the Company will cover the cost of your full COBRA premiums to continue your coverage (including coverage for eligible dependents, if applicable) in effect for yourself (and your eligible dependents, if applicable) until the earliest of: (A) seven 7 months following the Separation Date; (B) the expiration of your eligibility for the continuation coverage under COBRA; or (C) the date when you become eligible for and covered by substantially equivalent health insurance coverage in connection with new employment or self-employment (such period from the termination date through the earliest of (A) through (C), the “COBRA Payment Period” and such severance benefit, the “COBRA Severance Benefit”). Notwithstanding the foregoing, if at any time, (i) the Company determines, in its sole discretion, that the payment of the COBRA premiums would result in a violation of the nondiscrimination rules of Section 105(h)(2) of the Internal Revenue Code or any statute or regulation of similar effect (including but not limited to the 2010 Patient Protection and Affordable Care Act, as amended by the 2010 Health Care and Education Reconciliation Act), or (ii) the Company’s health insurance plan in effect on the Separation Date terminates (either (i) or (ii), a “COBRA Severance Benefit Terminating Event”), then provided you otherwise were eligible for the COBRA Severance Benefit on the date of the COBRA Severance Benefit Terminating Event, in lieu of providing the COBRA premium reimbursement, the Company will instead pay you a fully taxable lump sum cash payment equal to the remainder of the COBRA premiums due under this Section 3(b) for the COBRA Payment Period (the “Special Cash Payment”), which payment shall be payable within 30 days following the applicable COBRA Severance Benefit Terminating Event. You may, but are not obligated to, use such Special Cash Payment for medical expenses, including COBRA Premiums (as applicable). If, prior to any COBRA Severance Benefit Terminating Event, you become eligible for coverage under another employer’s group health plan through self-employment or otherwise cease to be eligible for COBRA coverage under the Company’s group health plan during the COBRA Payment Period, you must immediately notify the Company of such event, and all payments and obligations under this Section will cease.
(c) Consulting Agreement. The Company will continue to retain you in a consulting role pursuant to the Consulting Agreement attached as Exhibit A (the “Consulting Agreement”), which shall be deemed effective as of the Separation Date. Notwithstanding the foregoing, if you do not timely sign and return this Agreement, or you sign but later revoke your acceptance of this Agreement, the Consulting Agreement will terminate in accordance with its terms.
(d) Retention of Laptop Computer. As an additional Severance Benefit, the Company will permit you to retain the Company-issued laptop computer provided to you during your employment; provided, however, that you agree to reasonably cooperate with the Company to confirm all files stored on the computer are intact and all Company confidential information and trade secrets have been deleted after the end of the Consulting Period (as defined in the Consulting Agreement).
(e) Letter of Reference. As an additional Severance Benefit, upon your request, the Company will provide you with a signed letter of reference. This letter of reference will be
provided to you after you send a written request for the letter to the Company, provided you have returned this executed Agreement to the Company by the date of such request.
The Company is offering severance to you in reliance on Treasury Regulation Section 1.409A-1(b)(9) and the short-term deferral exception in Treasury Regulation Section 1.409A-1(b)(4). Any payments made in reliance on Treasury Regulation Section 1.409A-1(b)(4) will be made not later than March 15 of the year following the year in which the Separation Date occurs. For purposes of Section 409A of the Internal Revenue Code of 1986, as amended (“Code Section 409A”), your right to receive any installment payments under this letter (whether severance payments, reimbursements or otherwise) shall be treated as a right to receive a series of separate payments and, accordingly, each installment payment hereunder shall at all times be considered a separate and distinct payment. To the extent that any Severance Benefits are not otherwise exempt from the application of Code Section 409A, then, if the period during which you may consider and sign this Agreement spans two calendar years, the payment of severance will not be made or begin until the later calendar year.
4.Benefit Plans. If you are currently participating in the Company’s group health insurance plans, including medical, dental, and/or vision plans, your participation as an employee will end on last day of the month in which the Separation Date occurred. Thereafter, to the extent provided by the federal COBRA law or, if applicable, state insurance laws, and by the Company’s current group health insurance policies, you will be eligible to continue your group health insurance benefits at your own expense, with the potential for certain payments to be made by the Company as described in Section 3(b) above. For questions on your benefits, you may reach out to TriNet Solution Center at 800-638-0461 or employees@TriNet.com.
5.Unemployment Insurance. You may be eligible for unemployment insurance benefits after the Separation Date. You acknowledge that whether you receive unemployment compensation will be decided by the applicable agency that is charged with unemployment insurance matters in your state, and not by the Company. That agency can provide you with benefits and eligibility information regarding unemployment compensation.
6.Equity Awards. If you were granted stock options to purchase certain shares of the Company’s common stock (the “Options”) and/or restricted stock units to be issued shares of the Company’s common stock (the “RSUs” and together with the Options, the “Equity Awards”) pursuant to the Company’s 2016 Equity Incentive Plan (the “Plan”) and your applicable Option and/or RSU agreement (together with the Plan, the “Equity Award Documents”), vesting of your Options and/or RSUs (as applicable) will cease as of the date your “Continuous Service” (as defined in the Plan) ends. For avoidance of doubt, if you satisfy the Severance Preconditions, then the Equity Awards remain and will continue to remain outstanding and the unvested shares subject to the Equity Awards will continue to be eligible to vest following the Separation Date during the Consulting Period (as defined in the Consulting Agreement), in accordance with the vesting schedules applicable to such Equity Awards and subject to your Continuous Service during such periods. Any Options or RSUs that you currently hold shall continue to be governed by the terms of the Equity Award Documents.
7.Other Compensation or Benefits. You acknowledge that, except as expressly provided in this Agreement and the Consulting Agreement, you will not receive any additional
compensation, severance, commissions, bonuses or benefits after the Separation Date. You also acknowledge that the Company’s provision of the Severance Benefits set forth in Section 3 above fully satisfy any severance or other obligations under the Employment Agreement.
8.Expense Reimbursements. Within ten (10) days following the Separation Date, you agree to submit your final documented expense reimbursement statement reflecting all business expenses you incurred through the Separation Date, if any, for which you seek reimbursement. The Company will reimburse you for reasonable business expenses pursuant to its regular business practice.
9.Return of Company Property. Within three (3) days following the end of the Consulting Period, you agree to return to the Company all Company documents (and all copies thereof) and other Company property that you have had in your possession at any time, including, but not limited to, Company files, notes, drawings, records, business plans and forecasts, financial information, specifications, computer-recorded information, tangible property (other than the laptop referenced in Section 3 above), credit cards, entry cards, identification badges and keys; and, any materials of any kind that contain or embody any proprietary or confidential information of the Company (and all reproductions thereof).
10.Proprietary Information and Post-Termination Obligations. Both during and after your employment you acknowledge your continuing obligations under your Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement or any other confidentiality agreement you signed with the Company (the “Confidential Information Agreement”) not to use or disclose any confidential or proprietary information of the Company and to refrain from certain other activities. A copy of the Confidential Information Agreement is attached hereto as Exhibit B. As you know, the Company will enforce its contract rights. Please familiarize yourself with the enclosed agreement which you signed. Confidential information that is also a “trade secret,” as defined by law, may be disclosed (A) if it is made (i) in confidence to a federal, state, or local government official, either directly or indirectly, or to an attorney and (ii) solely for the purpose of reporting or investigating a suspected violation of law; or (B) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. In addition, in the event that you file a lawsuit for retaliation by the Company for reporting a suspected violation of law, you may disclose the trade secret to your attorney and use the trade secret information in the court proceeding, if you: (A) file any document containing the trade secret under seal; and (B) do not disclose the trade secret, except pursuant to court order.
11. Mutual Non-Disparagement. Both you and the Company agree not to disparage the other party, and the other party’s officers, directors, employees, shareholders and agents, in any manner reasonably likely to be harmful to them or their business, business reputation or personal reputation. The Company’s obligation not to disparage you under this Section are limited to the Company’s current officers and directors. Notwithstanding the foregoing, nothing in this Agreement shall limit your right to voluntarily communicate with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, other federal government agency or similar state or local agency or to discuss the terms and conditions of your employment with others to the extent
expressly permitted by Section 7 of the National Labor Relations Act. In addition, nothing in this Section or this Agreement is intended to prohibit or restrain you in any manner from making disclosures protected under the whistleblower provisions of federal or state law or regulation or other applicable law or regulation.
12.Cooperation after Termination. Consistent with your obligations under the Employment Agreement, you agree to cooperate fully with the Company in all matters relating to the transition of your work and responsibilities on behalf of the Company, including, but not limited to, any present, prior or subsequent relationships and the orderly transfer of any such work and institutional knowledge to such other persons as may be designated by the Company, by making yourself reasonably available during regular business hours.
(a)General Release of Claims. In exchange for the consideration provided to you under this Agreement to which you would not otherwise be entitled, you hereby generally and completely release the Company, and its affiliated, related, parent and subsidiary entities, and its and their current and former directors, officers, employees, shareholders, partners, agents, co-employers, including TriNet Group, Inc., attorneys, predecessors, successors, insurers, affiliates, and assigns (the “Company Parties”) from any and all claims, liabilities, demands, causes of action, and obligations, both known and unknown, arising from or in any way related to events, acts, conduct, or omissions occurring at any time prior to and including the date you sign this Agreement.
(b)Scope of Release. This general release includes, but is not limited to: (i) all claims arising from or in any way related to your employment with the Company or the termination of that employment; (ii) all claims related to your compensation or benefits from the Company, including salary, bonuses, commissions, vacation pay, expense reimbursements, severance pay, fringe benefits, stock, stock options, or any other ownership, equity, or profits interests in the Company; (iii) all claims for breach of contract, wrongful termination, and breach of the implied covenant of good faith and fair dealing; (iv) all tort claims, including claims for fraud, defamation, emotional distress, and discharge in violation of public policy; and (v) all federal, state, and local statutory claims, including claims for discrimination, harassment, retaliation, attorneys’ fees, or other claims arising under the federal Civil Rights Act of 1964 (as amended), the federal Americans with Disabilities Act of 1990, the Age Discrimination in Employment Act, as amended (“ADEA”), the Georgia Fair Employment Practices Act, the Georgia Equal Pay Act, the Georgia Age Discrimination in Employment Law, the Georgia Equal Employment for Persons with Disabilities Code, the Georgia Minimum Wage Law, the Georgia Equal Pay Act, the Georgia Law on Discrimination on the Basis of Maternity Leave, and the Georgia Law on Sex Discrimination.
(c)ADEA Release. You acknowledge that you are knowingly and voluntarily waiving and releasing any rights you have under the ADEA, and that the consideration given for the waiver and releases you have given in this Agreement is in addition to anything of value to which you were already entitled. You further acknowledge that you have been advised, as required by the ADEA, that: (i) your waiver and release does not apply to any rights or claims arising after the date you sign this Agreement; (ii) you should consult with an attorney prior to signing this Agreement (although you may choose voluntarily not to do so); (iii) you have forty-five (45) days
to consider this Agreement (although you may choose voluntarily to sign it sooner); (iv) you have seven (7) days following the date you sign this Agreement to revoke this Agreement (in a written revocation sent to the Company); and (v) this Agreement will not be effective until the date upon which the revocation period has expired, which will be the eighth day after you sign this Agreement provided that you do not revoke it. You also hereby acknowledge that the Company has provided you with the ADEA Disclosure Schedule (pursuant to Title 29 U.S. Code Section 626(f)(1)(H)), attached as Exhibit C to this Agreement.
(d)Exceptions. Notwithstanding the foregoing, you are not releasing the Company hereby from: (i) any claims that may arise from events that occur after the date this waiver is executed; (ii) any existing obligation to indemnify you pursuant to the Articles and Bylaws of the Company, any valid fully executed indemnification agreement with the Company, applicable law, or applicable directors and officers liability insurance; (iii) any claims that cannot be waived by law, including, without limitation, any rights you may have under applicable workers’ compensation laws and your right, if applicable, to file or participate in an investigative proceeding of any federal, state, or local government agency; or (iv) any claims for breach of this Agreement.
14.Protected Rights. You understand that nothing in this Agreement has limited, currently limits, or shall limit your ability to file a charge or complaint with the Equal Employment Opportunity Commission, the Department of Labor, the National Labor Relations Board, the Occupational Safety and Health Administration, the Department of Justice, the Securities and Exchange Commission, or any other government agency, law enforcement agency, or commission (“Government Agencies”). You further understand this Agreement does not limit your ability to communicate with any Government Agencies or otherwise participate in any investigation or proceeding that may be conducted by any Government Agency, including providing documents or other information, without notice to the Company. While this Agreement does not limit your right to receive a government-issued award for information provided to any Government Agency in connection with a government whistleblower program or protected whistleblower activity, you understand and agree that, to maximum extent permitted by law, you are otherwise waiving any and all rights you may have to individual relief based on any claims that you have released and any rights you have waived by signing this Agreement. Nothing in this Agreement has prevented, currently prevents, or shall prevent you from discussing or disclosing information about unlawful acts in the workplace, such as harassment, or discrimination or any other conduct that you have reason to believe is unlawful. Additionally, nothing in this Agreement waives any rights you may have under Section 7 of the National Labor Relations Act (subject to the release of claims set forth herein).
15.Your Acknowledgments and Affirmations. You acknowledge and agree that (i) the consideration given to you in exchange for the waiver and release in this Agreement is in addition to anything of value to which you were already entitled; and (ii) that you have been paid for all time worked, have received all the leave, leaves of absence and leave benefits and protections for which you are eligible, and have not suffered any on-the-job injury for which you have not already filed a claim. You affirm that all of the decisions of the Company Parties regarding your pay and benefits through the date of your execution of this Agreement were not discriminatory based on age, disability, race, color, sex, religion, national origin or any other classification protected by law. You affirm that you will not voluntarily (except in response to
legal compulsion or as permitted in Section 15 above) assist any person in bringing or pursuing any proposed or pending litigation, arbitration, administrative claim or other formal proceeding against any of the Company Parties. You further affirm that you have no known workplace injuries or occupational diseases. You acknowledge and affirm that you have not been retaliated against for reporting any allegation of corporate fraud or other wrongdoing by any of the Company Parties, or for exercising any rights protected by law, including any rights protected by the Fair Labor Standards Act, the Family Medical Leave Act, or any related statute or local leave or disability accommodation laws, or any applicable state workers’ compensation law.
16.No Admission. This Agreement does not constitute an admission by the Company of any wrongful action or violation of any federal, state, or local statute, or common law rights, including those relating to the provisions of any law or statute concerning employment actions, or of any other possible or claimed violation of law or rights.
17.Breach. You agree that upon any breach of this Agreement you will forfeit all amounts paid or owing to you under this Agreement. Further, you acknowledge that it may be impossible to assess the damages caused by your violation of the terms of Sections 9, 10, 11, and 12 of this Agreement and further agree that any threatened or actual violation or breach of those Sections of this Agreement will constitute immediate and irreparable injury to the Company. You therefore agree that any such breach of this Agreement is a material breach of this Agreement, and, in addition to any and all other damages and remedies available to the Company upon your breach of this Agreement, the Company shall be entitled to an injunction to prevent you from violating or breaching this Agreement. You agree that if the Company is successful in whole or part in any legal or equitable action against you under this Agreement, you agree to pay all of the costs, including reasonable attorneys’ fees, incurred by the Company in enforcing the terms of this Agreement.
18.Dispute Resolution. You and the Company agree that any and all disputes, claims, or controversies of any nature whatsoever arising from, or relating to, this Agreement or its interpretation, enforcement, breach, performance, or execution, or any of the matters herein released (collectively, “Claims,” each a “Claim”), shall be resolved, pursuant to the Federal Arbitration Act, 9 U.S.C. §1-16, and to the fullest extent permitted by law, by final, binding, and confidential arbitration at a location closest to where you last worked for the Company or another mutually agreeable location. The arbitration shall be conducted before a single neutral arbitrator by JAMS, Inc. (“JAMS”) or its successor, under the then applicable JAMS Comprehensive Arbitration Rules and Procedures (currently available at https://www.jamsadr.com/rules-comprehensive-arbitration) (“JAMS Rules”) and Georgia law. Both you and the Company opt into the Expedited Procedures under the JAMS Rules. The arbitrator shall apply substantive and procedural Georgia law to any dispute or claim, without reference to any conflict-of-law provisions of any jurisdiction. To the extent that the JAMS Rules conflict with Georgia law, Georgia law shall take precedence. The parties agree that punitive damages shall not be available in arbitration. By agreeing to this arbitration procedure, both you and the Company waive the right to have any Claim resolved through a trial by jury or judge or an administrative proceeding. This Section shall not apply to any action or claim that cannot be subject to mandatory arbitration as a matter of law. The arbitrator shall have sole authority for determining if a Claim is subject to arbitration, and any other procedural questions related to the dispute and bearing on the final disposition. In addition, the arbitrator shall: (a) have the authority to compel adequate discovery
for the resolution of the dispute and to award such relief as would otherwise be available under applicable law in a court proceeding; and (b) issue a written statement signed by the arbitrator regarding the disposition of each claim and the relief, if any, awarded as to each claim, the reasons for the award, and the arbitrator’s essential findings and conclusions on which the award is based. You and the Company shall each pay half the costs and expenses of the arbitration and each pay for its respective attorneys’ fees and costs, except as prohibited by law. To the extent JAMS does not collect or you otherwise do not pay to JAMS an equal share of all JAMS’ arbitration fees for any reason, and the Company pays JAMS your share, you acknowledge and agree that the Company shall be entitled to recover from you half of the JAMS arbitration fees invoiced to the parties (less any amounts you paid to JAMS) in a federal or state court of competent jurisdiction. Nothing in this letter agreement is intended to prevent either you or the Company from obtaining injunctive relief in court to prevent irreparable harm pending the conclusion of any such arbitration. Any awards or orders in such arbitrations may be entered and enforced as judgments in the federal and state courts of any competent jurisdiction.
_____ By initialing here, YOU acknowledge YOU have read and agree to this arbitration provision.
19.Miscellaneous. This Agreement, including Exhibits A, B and C, constitutes the complete, final and exclusive embodiment of the entire agreement between you and the Company with regard to this subject matter. It is entered into without reliance on any promise or representation, written or oral, other than those expressly contained herein, and it supersedes any other such promises, warranties or representations. This Agreement may not be modified or amended except in a writing signed by both you and a duly authorized officer of the Company. This Agreement will bind the heirs, personal representatives, successors and assigns of both you and the Company, and inure to the benefit of both you and the Company, their heirs, successors and assigns. If any provision of this Agreement is determined to be invalid or unenforceable, in whole or in part, this determination will not affect any other provision of this Agreement and the provision in question will be modified by the court so as to be rendered enforceable. This Agreement will be deemed to have been entered into and will be construed and enforced in accordance with the laws of the state or commonwealth in which you primarily performed work for the Company as applied to contracts made and to be performed entirely within such state or commonwealth.
If this Agreement is acceptable to you, please sign below and return it to me on or before the date that is forty-five (45) days after you receive this Agreement (but no earlier than the Separation Date). The Company’s severance offer contained herein will automatically expire if you do not sign and return the fully signed Agreement within this timeframe.
[SIGNATURES TO FOLLOW ON NEXT PAGE]
Sincerely,
Clearside Biomedical, Inc.
By: /s/George Lasezkay
George Lasezkay
President and Chief Executive Officer
I have read, understand and agree fully to the foregoing Agreement. I understand that this Agreement includes a release of all known and unknown claims, even those unknown claims that, if known by me, would affect my decision to accept this Agreement.
/s/Charlie Deignan
Charlie Deignan
July 19, 2025
Date
Exhibit A – Consulting Agreement
Exhibit B – Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C – ADEA disclosure
Exhibit A
Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and Charlie Deignan, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services as Principal Financial Officer and Principal Accounting Officer, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $304 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief
itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $633,525, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or
any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to
any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade
secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of
Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: ____________________________________
George Lasezkay
President and Chief Executive Officer
Agreed to and Accepted:
________________________________________
Charlie Deignan
____________________
Date
Exhibit B
Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C
ADEA Disclosure Schedule
The following disclosure was prepared as of July 17, 2025. It shows the employees in the decisional unit whose employment will be terminated pursuant to the business reorganization announced on or around July 17, 2025, and the factors used to determine who was eligible for the layoff. Employees who will be terminated will be eligible for benefits based upon their execution of a waiver. This data is subject to change and may be affected by future employment decisions.
Those receiving this disclosure will have forty-five (45) days to review the terms and conditions of the severance package.
On July 17, 2025, the employment of all individuals employed by the Company will be terminated in the business reorganization and no individuals employed by the Company will be retained.
The factors used to determine eligibility for the layoff are:
The Company’s assessment of its current and projected business and cost-reduction needs to effectuate its plan to cease all its active development activities and assess and execute strategic alternatives to its current business model. Not all criteria were used to make every selection.
EX-10.4

Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and Charlie Deignan, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services as Principal Financial Officer and Principal Accounting Officer, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $304 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $633,525, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable
time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall
be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such
services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local
agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation
Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to
Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: /s/George Lasezkay
George Lasezkay
President and Chief Executive Officer
Agreed to and Accepted:
/s/Charlie Deignan
Charlie Deignan
July 19, 2025
Date
EX-10.5

July 17, 2025
Via Email
Victor Chong
Re: Separation Agreement
Dear Victor:
This letter sets forth the substance of the separation agreement (the “Agreement”) which Clearside Biomedical, Inc. (the “Company”) is offering to you to aid in your employment transition.
1.Separation Date. Your last day of work with the Company and your employment termination date will be July 18, 2025 (the “Separation Date”). Between the date of this Agreement and your Separation Date, you will be expected to perform your regular job duties and such other duties as may be assigned to you by the Company’s Board of Directors (the “Board”). Notwithstanding anything to the contrary in Section 5(e) of your Employment Agreement (as defined below), you may continue your service as a director on the Board following your Separation Date.
2.Accrued Salary. In accordance with applicable law, the Company will pay you all accrued salary earned through the Separation Date subject to standard payroll deductions and withholdings. You acknowledge that you will receive these payments regardless of whether or not you execute this Agreement.
3.Severance Benefits. In lieu of the severance benefits you are eligible to receive pursuant to Section 5 of your June 19, 2024 Amended and Restated Executive Employment Agreement between you and the Company (the “Employment Agreement”), and provided you return this fully signed and dated Agreement to the Company within the time frame specified below (but no earlier than the Separation Date) do not revoke it, and fully comply with your obligations under this Agreement, and under your previously executed Confidential Information Agreement (as defined below) (collectively, the “Severance Preconditions”), the Company will, provide you with the following severance benefits (the “Severance Benefits”):
(a) Severance. The Company will pay you, as severance, the equivalent of twenty-six (26) weeks of your base salary in effect as of the Separation Date, subject to standard payroll deductions and withholdings. This amount will be paid in a lump sum on the Company’s next
regular payroll date following the Effective Date (as defined below), provided you have signed and not revoked the Agreement by such date.
(b) COBRA Severance Benefit. If you timely elect continued coverage under COBRA under the Company’s group health plans, then, as an additional severance benefit, the Company will cover the cost of your full COBRA premiums to continue your coverage (including coverage for eligible dependents, if applicable) in effect for yourself (and your eligible dependents, if applicable) until the earliest of: (A) Six (6) months following the Separation Date; (B) the expiration of your eligibility for the continuation coverage under COBRA; or (C) the date when you become eligible for and covered by substantially equivalent health insurance coverage in connection with new employment or self-employment (such period from the termination date through the earliest of (A) through (C), the “COBRA Payment Period” and such severance benefit, the “COBRA Severance Benefit”). Notwithstanding the foregoing, if at any time, (i) the Company determines, in its sole discretion, that the payment of the COBRA premiums would result in a violation of the nondiscrimination rules of Section 105(h)(2) of the Internal Revenue Code or any statute or regulation of similar effect (including but not limited to the 2010 Patient Protection and Affordable Care Act, as amended by the 2010 Health Care and Education Reconciliation Act), or (ii) the Company’s health insurance plan in effect on the Separation Date terminates (either (i) or (ii), a “COBRA Severance Benefit Terminating Event”), then provided you otherwise were eligible for the COBRA Severance Benefit on the date of the COBRA Severance Benefit Terminating Event, in lieu of providing the COBRA premium reimbursement, the Company will instead pay you a fully taxable lump sum cash payment equal to the remainder of the COBRA premiums due under this Section 3(b) for the COBRA Payment Period (the “Special Cash Payment”), which payment shall be payable within 30 days following the applicable COBRA Severance Benefit Terminating Event. You may, but are not obligated to, use such Special Cash Payment for medical expenses, including COBRA Premiums (as applicable). If, prior to any COBRA Severance Benefit Terminating Event, you become eligible for coverage under another employer’s group health plan through self-employment or otherwise cease to be eligible for COBRA coverage under the Company’s group health plan during the COBRA Payment Period, you must immediately notify the Company of such event, and all payments and obligations under this Section will cease.
(c) Consulting Agreement. The Company will continue to retain you in a consulting role pursuant to the Consulting Agreement attached as Exhibit A (the “Consulting Agreement”), which shall be deemed effective as of the Separation Date. Notwithstanding the foregoing, if you do not timely sign and return this Agreement, or you sign but later revoke your acceptance of this Agreement, the Consulting Agreement will terminate in accordance with its terms.
(d) Retention of Laptop Computer. As an additional Severance Benefit, the Company will permit you to retain the Company-issued laptop computer provided to you during your employment; provided, however, that you agree to reasonably cooperate with the Company to confirm all files stored on the computer are intact and all Company confidential information and trade secrets have been deleted after the end of the Consulting Period (as defined in the Consulting Agreement).
(e) Letter of Reference. As an additional Severance Benefit, upon your request, the Company will provide you with a signed letter of reference. This letter of reference will be
provided to you after you send a written request for the letter to the Company, provided you have returned this executed Agreement to the Company by the date of such request.
The Company is offering severance to you in reliance on Treasury Regulation Section 1.409A-1(b)(9) and the short-term deferral exception in Treasury Regulation Section 1.409A-1(b)(4). Any payments made in reliance on Treasury Regulation Section 1.409A-1(b)(4) will be made not later than March 15 of the year following the year in which the Separation Date occurs. For purposes of Section 409A of the Internal Revenue Code of 1986, as amended (“Code Section 409A”), your right to receive any installment payments under this letter (whether severance payments, reimbursements or otherwise) shall be treated as a right to receive a series of separate payments and, accordingly, each installment payment hereunder shall at all times be considered a separate and distinct payment. To the extent that any Severance Benefits are not otherwise exempt from the application of Code Section 409A, then, if the period during which you may consider and sign this Agreement spans two calendar years, the payment of severance will not be made or begin until the later calendar year.
4.Benefit Plans. If you are currently participating in the Company’s group health insurance plans, including medical, dental, and/or vision plans, your participation as an employee will end on last day of the month in which the Separation Date occurred. Thereafter, to the extent provided by the federal COBRA law or, if applicable, state insurance laws, and by the Company’s current group health insurance policies, you will be eligible to continue your group health insurance benefits at your own expense, with the potential for certain payments to be made by the Company as described in Section 3(b) above. For questions on your benefits, you may reach out to TriNet Solution Center at 800-638-0461 or employees@TriNet.com.
5.Unemployment Insurance. You may be eligible for unemployment insurance benefits after the Separation Date. You acknowledge that whether you receive unemployment compensation will be decided by the applicable agency that is charged with unemployment insurance matters in your state, and not by the Company. That agency can provide you with benefits and eligibility information regarding unemployment compensation.
6.Equity Awards. If you were granted stock options to purchase certain shares of the Company’s common stock (the “Options”) and/or restricted stock units to be issued shares of the Company’s common stock (the “RSUs” and together with the Options, the “Equity Awards”) pursuant to the Company’s 2016 Equity Incentive Plan (the “Plan”) and your applicable Option and/or RSU agreement (together with the Plan, the “Equity Award Documents”), vesting of your Options and/or RSUs (as applicable) will cease as of the date your “Continuous Service” (as defined in the Plan) ends. For avoidance of doubt, if you satisfy the Severance Preconditions, then the Equity Awards remain and will continue to remain outstanding and the unvested shares subject to the Equity Awards will continue to be eligible to vest following the Separation Date during the Consulting Period (as defined in the Consulting Agreement), in accordance with the vesting schedules applicable to such Equity Awards and subject to your Continuous Service during such periods. Any Options or RSUs that you currently hold shall continue to be governed by the terms of the Equity Award Documents.
7.Other Compensation or Benefits. You acknowledge that, except as expressly provided in this Agreement and the Consulting Agreement, you will not receive any additional
compensation, severance, commissions, bonuses or benefits after the Separation Date. You also acknowledge that the Company’s provision of the Severance Benefits set forth in Section 3 above fully satisfy any severance or other obligations under the Employment Agreement.
8.Expense Reimbursements. Within ten (10) days following the Separation Date, you agree to submit your final documented expense reimbursement statement reflecting all business expenses you incurred through the Separation Date, if any, for which you seek reimbursement. The Company will reimburse you for reasonable business expenses pursuant to its regular business practice.
9.Return of Company Property. Within three (3) days following the end of the Consulting Period, you agree to return to the Company all Company documents (and all copies thereof) and other Company property that you have had in your possession at any time, including, but not limited to, Company files, notes, drawings, records, business plans and forecasts, financial information, specifications, computer-recorded information, tangible property (other than the laptop referenced in Section 3 above), credit cards, entry cards, identification badges and keys; and, any materials of any kind that contain or embody any proprietary or confidential information of the Company (and all reproductions thereof).
10.Proprietary Information and Post-Termination Obligations. Both during and after your employment you acknowledge your continuing obligations under your Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement or any other confidentiality agreement you signed with the Company (the “Confidential Information Agreement”) not to use or disclose any confidential or proprietary information of the Company and to refrain from certain other activities. A copy of the Confidential Information Agreement is attached hereto as Exhibit B. As you know, the Company will enforce its contract rights. Please familiarize yourself with the enclosed agreement which you signed. Confidential information that is also a “trade secret,” as defined by law, may be disclosed (A) if it is made (i) in confidence to a federal, state, or local government official, either directly or indirectly, or to an attorney and (ii) solely for the purpose of reporting or investigating a suspected violation of law; or (B) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. In addition, in the event that you file a lawsuit for retaliation by the Company for reporting a suspected violation of law, you may disclose the trade secret to your attorney and use the trade secret information in the court proceeding, if you: (A) file any document containing the trade secret under seal; and (B) do not disclose the trade secret, except pursuant to court order.
11. Mutual Non-Disparagement. Both you and the Company agree not to disparage the other party, and the other party’s officers, directors, employees, shareholders and agents, in any manner reasonably likely to be harmful to them or their business, business reputation or personal reputation. The Company’s obligation not to disparage you under this Section are limited to the Company’s current officers and directors. Notwithstanding the foregoing, nothing in this Agreement shall limit your right to voluntarily communicate with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, other federal government agency or similar state or local agency or to discuss the terms and conditions of your employment with others to the extent expressly permitted by Section 7 of the National Labor Relations Act. In addition, nothing in this Section or this Agreement is intended to prohibit or restrain you in any manner from making
disclosures protected under the whistleblower provisions of federal or state law or regulation or other applicable law or regulation.
12.Cooperation after Termination. Consistent with your obligations under the Employment Agreement, you agree to cooperate fully with the Company in all matters relating to the transition of your work and responsibilities on behalf of the Company, including, but not limited to, any present, prior or subsequent relationships and the orderly transfer of any such work and institutional knowledge to such other persons as may be designated by the Company, by making yourself reasonably available during regular business hours.
(a)General Release of Claims. In exchange for the consideration provided to you under this Agreement to which you would not otherwise be entitled, you hereby generally and completely release the Company, and its affiliated, related, parent and subsidiary entities, and its and their current and former directors, officers, employees, shareholders, partners, agents, co-employers, including TriNet Group, Inc., attorneys, predecessors, successors, insurers, affiliates, and assigns (the “Company Parties”) from any and all claims, liabilities, demands, causes of action, and obligations, both known and unknown, arising from or in any way related to events, acts, conduct, or omissions occurring at any time prior to and including the date you sign this Agreement.
(b)Scope of Release. This general release includes, but is not limited to: (i) all claims arising from or in any way related to your employment with the Company or the termination of that employment; (ii) all claims related to your compensation or benefits from the Company, including salary, bonuses, commissions, vacation pay, expense reimbursements, severance pay, fringe benefits, stock, stock options, or any other ownership, equity, or profits interests in the Company; (iii) all claims for breach of contract, wrongful termination, and breach of the implied covenant of good faith and fair dealing; (iv) all tort claims, including claims for fraud, defamation, emotional distress, and discharge in violation of public policy; and (v) all federal, state, and local statutory claims, including claims for discrimination, harassment, retaliation, attorneys’ fees, or other claims arising under the federal Civil Rights Act of 1964 (as amended), the federal Americans with Disabilities Act of 1990, the Age Discrimination in Employment Act, as amended (“ADEA”), the Georgia Fair Employment Practices Act, the Georgia Equal Pay Act, the Georgia Age Discrimination in Employment Law, the Georgia Equal Employment for Persons with Disabilities Code, the Georgia Minimum Wage Law, the Georgia Equal Pay Act, the Georgia Law on Discrimination on the Basis of Maternity Leave, and the Georgia Law on Sex Discrimination.
(c)ADEA Release. You acknowledge that you are knowingly and voluntarily waiving and releasing any rights you have under the ADEA, and that the consideration given for the waiver and releases you have given in this Agreement is in addition to anything of value to which you were already entitled. You further acknowledge that you have been advised, as required by the ADEA, that: (i) your waiver and release does not apply to any rights or claims arising after the date you sign this Agreement; (ii) you should consult with an attorney prior to signing this Agreement (although you may choose voluntarily not to do so); (iii) you have forty-five (45) days to consider this Agreement (although you may choose voluntarily to sign it sooner); (iv) you have seven (7) days following the date you sign this Agreement to revoke this Agreement (in a written
revocation sent to the Company); and (v) this Agreement will not be effective until the date upon which the revocation period has expired, which will be the eighth day after you sign this Agreement provided that you do not revoke it. You also hereby acknowledge that the Company has provided you with the ADEA Disclosure Schedule (pursuant to Title 29 U.S. Code Section 626(f)(1)(H)), attached as Exhibit C to this Agreement.
(d)Exceptions. Notwithstanding the foregoing, you are not releasing the Company hereby from: (i) any claims that may arise from events that occur after the date this waiver is executed; (ii) any existing obligation to indemnify you pursuant to the Articles and Bylaws of the Company, any valid fully executed indemnification agreement with the Company, applicable law, or applicable directors and officers liability insurance; (iii) any claims that cannot be waived by law, including, without limitation, any rights you may have under applicable workers’ compensation laws and your right, if applicable, to file or participate in an investigative proceeding of any federal, state, or local government agency; or (iv) any claims for breach of this Agreement.
14.Protected Rights. You understand that nothing in this Agreement has limited, currently limits, or shall limit your ability to file a charge or complaint with the Equal Employment Opportunity Commission, the Department of Labor, the National Labor Relations Board, the Occupational Safety and Health Administration, the Department of Justice, the Securities and Exchange Commission, or any other government agency, law enforcement agency, or commission (“Government Agencies”). You further understand this Agreement does not limit your ability to communicate with any Government Agencies or otherwise participate in any investigation or proceeding that may be conducted by any Government Agency, including providing documents or other information, without notice to the Company. While this Agreement does not limit your right to receive a government-issued award for information provided to any Government Agency in connection with a government whistleblower program or protected whistleblower activity, you understand and agree that, to maximum extent permitted by law, you are otherwise waiving any and all rights you may have to individual relief based on any claims that you have released and any rights you have waived by signing this Agreement. Nothing in this Agreement has prevented, currently prevents, or shall prevent you from discussing or disclosing information about unlawful acts in the workplace, such as harassment, or discrimination or any other conduct that you have reason to believe is unlawful. Additionally, nothing in this Agreement waives any rights you may have under Section 7 of the National Labor Relations Act (subject to the release of claims set forth herein).
15.Your Acknowledgments and Affirmations. You acknowledge and agree that (i) the consideration given to you in exchange for the waiver and release in this Agreement is in addition to anything of value to which you were already entitled; and (ii) that you have been paid for all time worked, have received all the leave, leaves of absence and leave benefits and protections for which you are eligible, and have not suffered any on-the-job injury for which you have not already filed a claim. You affirm that all of the decisions of the Company Parties regarding your pay and benefits through the date of your execution of this Agreement were not discriminatory based on age, disability, race, color, sex, religion, national origin or any other classification protected by law. You affirm that you will not voluntarily (except in response to legal compulsion or as permitted in Section 15 above) assist any person in bringing or pursuing any proposed or pending litigation, arbitration, administrative claim or other formal proceeding
against any of the Company Parties. You further affirm that you have no known workplace injuries or occupational diseases. You acknowledge and affirm that you have not been retaliated against for reporting any allegation of corporate fraud or other wrongdoing by any of the Company Parties, or for exercising any rights protected by law, including any rights protected by the Fair Labor Standards Act, the Family Medical Leave Act, or any related statute or local leave or disability accommodation laws, or any applicable state workers’ compensation law.
16.No Admission. This Agreement does not constitute an admission by the Company of any wrongful action or violation of any federal, state, or local statute, or common law rights, including those relating to the provisions of any law or statute concerning employment actions, or of any other possible or claimed violation of law or rights.
17.Breach. You agree that upon any breach of this Agreement you will forfeit all amounts paid or owing to you under this Agreement. Further, you acknowledge that it may be impossible to assess the damages caused by your violation of the terms of Sections 9, 10, 11, and 12 of this Agreement and further agree that any threatened or actual violation or breach of those Sections of this Agreement will constitute immediate and irreparable injury to the Company. You therefore agree that any such breach of this Agreement is a material breach of this Agreement, and, in addition to any and all other damages and remedies available to the Company upon your breach of this Agreement, the Company shall be entitled to an injunction to prevent you from violating or breaching this Agreement. You agree that if the Company is successful in whole or part in any legal or equitable action against you under this Agreement, you agree to pay all of the costs, including reasonable attorneys’ fees, incurred by the Company in enforcing the terms of this Agreement.
18.Dispute Resolution. You and the Company agree that any and all disputes, claims, or controversies of any nature whatsoever arising from, or relating to, this Agreement or its interpretation, enforcement, breach, performance, or execution, or any of the matters herein released (collectively, “Claims,” each a “Claim”), shall be resolved, pursuant to the Federal Arbitration Act, 9 U.S.C. §1-16, and to the fullest extent permitted by law, by final, binding, and confidential arbitration at a location closest to where you last worked for the Company or another mutually agreeable location. The arbitration shall be conducted before a single neutral arbitrator by JAMS, Inc. (“JAMS”) or its successor, under the then applicable JAMS Comprehensive Arbitration Rules and Procedures (currently available at https://www.jamsadr.com/rules-comprehensive-arbitration) (“JAMS Rules”) and Georgia law. Both you and the Company opt into the Expedited Procedures under the JAMS Rules. The arbitrator shall apply substantive and procedural Georgia law to any dispute or claim, without reference to any conflict-of-law provisions of any jurisdiction. To the extent that the JAMS Rules conflict with Georgia law, Georgia law shall take precedence. The parties agree that punitive damages shall not be available in arbitration. By agreeing to this arbitration procedure, both you and the Company waive the right to have any Claim resolved through a trial by jury or judge or an administrative proceeding. This Section shall not apply to any action or claim that cannot be subject to mandatory arbitration as a matter of law. The arbitrator shall have sole authority for determining if a Claim is subject to arbitration, and any other procedural questions related to the dispute and bearing on the final disposition. In addition, the arbitrator shall: (a) have the authority to compel adequate discovery for the resolution of the dispute and to award such relief as would otherwise be available under applicable law in a court proceeding; and (b) issue a written statement signed by the arbitrator
regarding the disposition of each claim and the relief, if any, awarded as to each claim, the reasons for the award, and the arbitrator’s essential findings and conclusions on which the award is based. You and the Company shall each pay half the costs and expenses of the arbitration and each pay for its respective attorneys’ fees and costs, except as prohibited by law. To the extent JAMS does not collect or you otherwise do not pay to JAMS an equal share of all JAMS’ arbitration fees for any reason, and the Company pays JAMS your share, you acknowledge and agree that the Company shall be entitled to recover from you half of the JAMS arbitration fees invoiced to the parties (less any amounts you paid to JAMS) in a federal or state court of competent jurisdiction. Nothing in this letter agreement is intended to prevent either you or the Company from obtaining injunctive relief in court to prevent irreparable harm pending the conclusion of any such arbitration. Any awards or orders in such arbitrations may be entered and enforced as judgments in the federal and state courts of any competent jurisdiction.
_____ By initialing here, YOU acknowledge YOU have read and agree to this arbitration provision.
19.Miscellaneous. This Agreement, including Exhibits A, B and C, constitutes the complete, final and exclusive embodiment of the entire agreement between you and the Company with regard to this subject matter. It is entered into without reliance on any promise or representation, written or oral, other than those expressly contained herein, and it supersedes any other such promises, warranties or representations. This Agreement may not be modified or amended except in a writing signed by both you and a duly authorized officer of the Company. This Agreement will bind the heirs, personal representatives, successors and assigns of both you and the Company, and inure to the benefit of both you and the Company, their heirs, successors and assigns. If any provision of this Agreement is determined to be invalid or unenforceable, in whole or in part, this determination will not affect any other provision of this Agreement and the provision in question will be modified by the court so as to be rendered enforceable. This Agreement will be deemed to have been entered into and will be construed and enforced in accordance with the laws of the state or commonwealth in which you primarily performed work for the Company as applied to contracts made and to be performed entirely within such state or commonwealth.
If this Agreement is acceptable to you, please sign below and return it to me on or before the date that is forty-five (45) days after you receive this Agreement (but no earlier than the Separation Date). The Company’s severance offer contained herein will automatically expire if you do not sign and return the fully signed Agreement within this timeframe.
[SIGNATURES TO FOLLOW ON NEXT PAGE]
Sincerely,
Clearside Biomedical, Inc.
By: /s/George Lasezkay
George Lasezkay
President and Chief Executive Officer
I have read, understand and agree fully to the foregoing Agreement. I understand that this Agreement includes a release of all known and unknown claims, even those unknown claims that, if known by me, would affect my decision to accept this Agreement.
/s/Victor Chong
Victor Chong
July 18, 2025
Date
Exhibit A – Consulting Agreement
Exhibit B – Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C – ADEA disclosure
Exhibit A
Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and Victor Chong, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $341 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief
itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $661,500, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or
any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to
any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade
secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of
Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: ____________________________________
George Lasezkay
President and Chief Executive Officer
Agreed to and Accepted:
________________________________________
Victor Chong
____________________
Date
Exhibit B
Employee Confidential Information, Inventions, Non-Solicitation and Non-Competition Agreement
Exhibit C
ADEA Disclosure Schedule
The following disclosure was prepared as of July 17, 2025. It shows the employees in the decisional unit whose employment will be terminated pursuant to the business reorganization announced on or around July 17, 2025, and the factors used to determine who was eligible for the layoff. Employees who will be terminated will be eligible for benefits based upon their execution of a waiver. This data is subject to change and may be affected by future employment decisions.
Those receiving this disclosure will have forty-five (45) days to review the terms and conditions of the severance package.
On July 17, 2025, the employment of all individuals employed by the Company will be terminated in the business reorganization and no individuals employed by the Company will be retained.
The factors used to determine eligibility for the layoff are:
The Company’s assessment of its current and projected business and cost-reduction needs to effectuate its plan to cease all its active development activities and assess and execute strategic alternatives to its current business model. Not all criteria were used to make every selection.
EX-10.6

Consulting Agreement
This Consulting Agreement (the “Consulting Agreement”) by and between Clearside Biomedical, Inc. (“Client”) and Victor Chong, an individual (“Consultant”) is effective as of July 18, 2025 (the “Consulting Agreement Effective Date”), subject to the terms of Section 14 below.
RECITALS
WHEREAS the parties desire for the Client to engage Consultant to perform the services described herein and for Consultant to provide such services on the terms and conditions described herein; and
WHEREAS, the parties desire to use Consultant’s independent skill and expertise pursuant to this Agreement as an independent contractor;
NOW THEREFORE, in consideration of the promises and mutual agreements contained herein, the parties hereto, intending to be legally bound, agree as follows:
1.Engagement of Services. Consultant agrees to provide consulting services, under the direction of the Board or their designee (the “Board”) of the Client. Specifically, Consultant may be asked to provide services in the areas of: licensing/partner support, training, regulatory, safety data base, supply, manufacturing, QA, engineering, accounting and SEC reporting and may be asked to perform certain transition activities upon reasonable request of the Company’s Board. Consultant agrees to exercise the highest degree of professionalism and utilize his expertise and creative talents in performing these services. Consultant agrees to make himself or herself reasonably available to perform such consulting services throughout the Consulting Period (as defined in Section 14.1), and to be reasonably available to meet with the Client. The parties anticipate that Consultant’s services will not require more than 20 hours per week on average during the Consulting Period.
2.Compensation. In consideration for the services rendered pursuant to this Agreement and for the assignment of certain Consultant’s right, title and interest pursuant hereto,
(a) Consulting Fees. During the Consulting Period, Consultant will be paid an hourly consulting fee of $341 for each hour of services performed under this Agreement (the “Consulting Fee”).
(b) Expenses. Consultant will be eligible to receive reimbursements for preapproved expenses incurred in connection with the provision of Consultant’s services.
(c) Invoices. Consultant shall invoice Client within ten (10) days after the close of each month during the Consulting Period for services rendered and expenses incurred during the previous month. Such invoices shall list the dates covered by the invoice and the hours that Consultant performed services (including a brief itemized description of the services rendered by Consultant and expenses incurred). The Client shall pay net thirty (30) days from receipt of the invoice.
(d) Equity Awards. During the Consulting Period, Consultant will be deemed to be in Continuous Service for purposes of vesting of the Equity Awards (as defined in the “Separation Agreement” dated July 17, 2025, to which this Agreement is attached as Exhibit A). All matters of vesting and exercisability of Consultant’s Equity Awards shall be as governed by the Separation Agreement and the terms of the Equity Award Documents (each as defined in the Separation Agreement), except as may be modified in Section 2(e) herein.
(e) Completion Payment. In the event the Consultant is continuing to provide services under this Consulting Agreement through the consummation of a Change in Control or Corporate Transaction (each as defined in the Plan) of the Client, and provided that Consultant has not been given an offer of employment with any acquirer in such Change in Control or Corporate Transaction (as applicable) (the “Buyer”) on substantially similar terms to those set forth in Consultant’s prior Employment Agreement with Client (an “Offer”), then the Consultant will receive an additional payment (the “Completion Payment”) in the total amount of $661,500, subject to any applicable payroll withholdings and deductions and payable at the time of closing of the applicable Change in Control or Corporate Transaction. Additionally, and notwithstanding anything to the contrary in the Plan or Equity Award Documents, any then-unvested Equity Awards held by the Consultant as of the date of the consummation of such Change in Control or Corporate Transaction, as applicable, shall be deemed vested and exercisable as of such date. In the event the Consultant accepts an Offer but is then terminated without cause by the applicable Buyer within one year of the consummation of the Change in Control or Corporate Transaction, as applicable, then the Client shall make commercially reasonable efforts to make a Completion Payment to Consultant within thirty (30) days of such termination without cause, provided the Client has been given timely notice of such termination.
3.Ownership of Work Product. Consultant hereby irrevocably assigns, grants and conveys to Client all right, title and interest now existing or that may exist in the future in and to any document, development, work product, know-how, design, processes, invention, technique, trade secret, or idea, and all intellectual property rights related thereto, that is created by Consultant, to which Consultant contributes, or which relates to Consultant’s services provided pursuant to this Agreement (the “Work Product”), including all copyrights, trademarks and other intellectual property rights (including but not limited to patent rights) relating thereto. Consultant agrees that any and all Work Product shall be and remain the property of Client. Consultant will immediately disclose to the Client all Work Product. Consultant agrees to execute, at Client’s request and expense, all documents and other instruments necessary or desirable to confirm such assignment. In the event that Consultant does not, for any reason, execute such documents within a reasonable
time of Client’s request, Consultant hereby irrevocably appoints Client as Consultant’s attorney-in-fact for the purpose of executing such documents on Consultant’s behalf, which appointment is coupled with an interest. Consultant shall not attempt to register any works created by Consultant pursuant to this Agreement at the U.S. Copyright Office, the U.S. Patent & Trademark Office, or any foreign copyright, patent, or trademark registry. Consultant retains no rights in the Work Product and agrees not to challenge Client’s ownership of the rights embodied in the Work Product. Consultant further agrees to assist Client in every proper way to enforce Client’s rights relating to the Work Product in any and all countries, including, but not limited to, executing, verifying and delivering such documents and performing such other acts (including appearing as a witness) as Client may reasonably request for use in obtaining, perfecting, evidencing, sustaining and enforcing Client’s rights relating to the Work Product.
4.Artist’s, Moral, and Other Rights. If Consultant has any rights, including without limitation “artist’s rights” or “moral rights,” in the Work Product which cannot be assigned (the “Non-Assignable Rights”), Consultant agrees to waive enforcement worldwide of such rights against Client. In the event that Consultant has any such rights that cannot be assigned or waived Consultant hereby grants to Client a royalty-free, paid-up, exclusive, worldwide, irrevocable, perpetual license under the Non-Assignable Rights to (i) use, make, sell, offer to sell, have made, and further sublicense the Work Product, and (ii) reproduce, distribute, create derivative works of, publicly perform and publicly display the Work Product in any medium or format, whether now known or later developed.
5.Representations and Warranties. Consultant represents and warrants that: (a) Consultant has the full right and authority to enter into this Agreement and perform his obligations hereunder; (b) Consultant has the right and unrestricted ability to assign the Work Product to Client as set forth in Sections 3 and 4 (including without limitation the right to assign any Work Product created by Consultant’s employees or contractors); (c) the Work Product has not heretofore been published in its entirety; and (d) the Work Product will not infringe upon any copyright, patent, trademark, right of publicity or privacy, or any other proprietary right of any person, whether contractual, statutory or common law. Consultant agrees to indemnify Client from any and all damages, costs, claims, expenses or other liability (including reasonable attorneys’ fees) arising from or relating to the breach or alleged breach by Consultant of the representations and warranties set forth in this Section 5.
6.Independent Contractor Relationship. Consultant is an independent contractor and not an employee of the Client. Nothing in this Agreement is intended to, or should be construed to, create a partnership, agency, joint venture or employment relationship. The manner and means by which Consultant chooses to complete the consulting services are in Consultant’s sole discretion and control. In completing the consulting services, Consultant agrees to provide his own equipment, tools and other materials at his own expense. Consultant is not authorized to represent that he is an agent, employee, or legal representative of the Client. Consultant is not authorized to make any representation, contract, or commitment on behalf of Client or incur any liabilities or obligations of any kind in the name of or on behalf of the Client. Consultant shall be free at all times to arrange the time and manner of performance of the consulting services. Consultant is not required to maintain any schedule of duties or assignments. Consultant is also not required to provide reports to the Client. In addition to all other obligations contained herein, Consultant agrees: (a) to proceed with diligence and promptness and hereby warrants that such services shall
be performed in accordance with the highest professional standards in the field to the satisfaction of the Client; and (b) to comply, at Consultant’s own expense, with the provisions of all state, local, and federal laws, regulations, ordinances, requirements and codes which are applicable to the performance of the services hereunder.
7.Consultant’s Responsibilities. As an independent contractor, the mode, manner, method and means used by Consultant in the performance of services shall be of Consultant’s selection and under the sole control and direction of Consultant. Consultant shall be responsible for all risks incurred in the operation of Consultant’s business and shall enjoy all the benefits thereof. Any persons employed by or subcontracting with Consultant to perform any part of Consultant’s obligations hereunder shall be under the sole control and direction of Consultant and Consultant shall be solely responsible for all liabilities and expenses thereof. The Client shall have no right or authority with respect to the selection, control, direction, or compensation of such persons.
8.Tax Treatment. Consultant and the Client agree that (except as otherwise set forth below) the Client will treat Consultant as an independent contractor for purposes of all tax laws (local, state and federal) and file forms consistent with that status. Consultant agrees, as an independent contractor, that neither he nor his employees are entitled to unemployment benefits in the event this Agreement terminates, or workers’ compensation benefits in the event that Consultant, or any employee of Consultant, is injured in any manner while performing obligations under this Agreement.
9.No Employee Benefits. Except as otherwise described in the Separation Agreement, Consultant acknowledges and agrees that neither he nor anyone acting on his behalf shall receive any employee benefits of any kind from the Client. Consultant (and Consultant’s agents, employees, and subcontractors) is excluded from participating in any fringe benefit plans or programs as a result of the performance of services under this Agreement, without regard to Consultant’s independent contractor status. In addition, Consultant (on behalf of himself and on behalf of Consultant’s agents, employees, and contractors) waives any and all rights, if any, to participation in any of the Client’s fringe benefit plans or programs including, but not limited to, health, sickness, accident or dental coverage, life insurance, disability benefits, severance, accidental death and dismemberment coverage, unemployment insurance coverage, workers’ compensation coverage, and pension or 401(k) benefit(s) provided by the Client to its employees. Notwithstanding the above, this Agreement does not amend or abrogate in any manner any benefits owed to Consultant under any qualified retirement plan or health and welfare benefit plan in which Consultant was a participant during his previous employment relationship with the Client.
10.Expenses and Liabilities. Consultant agrees that as an independent contractor, he is solely responsible for all expenses (and profits/losses) he incurs in connection with the performance of services. Consultant understands that he will not be reimbursed for any supplies, equipment, or operating costs, nor will these costs of doing business be defrayed in any way by the Client. In addition, the Client does not guarantee to Consultant that fees derived from Consultant’s business will exceed Consultant’s costs.
11.Non-Exclusivity. The Client reserves the right to engage other consultants to perform services, without giving Consultant a right of first refusal or any other exclusive rights. Consultant reserves the right to perform services for other persons, provided that the performance of such
services do not conflict or interfere with services provided pursuant to or obligations under this Agreement.
12.No Conflict of Interest. During the term of this Agreement, unless written permission is given by the Board, Consultant will not accept work, enter into a contract, or provide services to any third party that provides products or services which compete with the products or services provided by the Client nor may Consultant enter into any agreement or perform any services which would conflict or interfere with the services provided pursuant to or the obligations under this Agreement. Consultant warrants that there is no other contract or duty on his part that prevents or impedes Consultant’s performance under this Agreement. Consultant agrees to indemnify Client from any and all loss or liability incurred by reason of the alleged breach by Consultant of any services agreement with any third party.
13.Confidential Information. Consultant agrees to hold Client’s Confidential Information (as defined below) in strict confidence and not to disclose such Confidential Information to any third parties. Consultant also agrees not to use any of Client’s Confidential Information for any purpose other than performance of Consultant’s services hereunder. “Confidential Information” as used in this Agreement shall mean all information disclosed by Client to Consultant, or otherwise, regarding Client or its business obtained by Consultant pursuant to services provided under this Agreement that is not generally known in the Client’s trade or industry and shall include, without limitation, (a) concepts and ideas relating to the development and distribution of content in any medium or to the current, future and proposed products or services of Client or its subsidiaries or affiliates; (b) trade secrets, drawings, inventions, know-how, software programs, and software source documents; (c) information regarding plans for research, development, new service offerings or products, marketing and selling, business plans, business forecasts, budgets and unpublished financial statements, licenses and distribution arrangements, prices and costs, suppliers and customers; and (d) any information regarding the skills and compensation of employees, contractors or other agents of the Client or its subsidiaries or affiliates. Confidential Information also includes proprietary or confidential information of any third party who may disclose such information to Client or Consultant in the course of Client’s business. Consultant’s obligations set forth in this Section shall not apply with respect to any portion of the Confidential Information that Consultant can document by competent proof that such portion: (i) is in the public domain through no fault of Consultant; (ii) has been rightfully independently communicated to Consultant free of any obligation of confidence; or (iii) was developed by Consultant independently of and without reference to any information communicated to Consultant by Client. In addition, Consultant may disclose Client’s Confidential Information in response to a valid order by a court or other governmental body, as otherwise required by law. All Confidential Information furnished to Consultant by Client is the sole and exclusive property of Client or its suppliers or customers. Upon request by Client, Consultant agrees to promptly deliver to Client the original and any copies of such Confidential Information. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and Client. Notwithstanding the foregoing or anything to the contrary in this Agreement or any other agreement between Client and Consultant, nothing in this Agreement shall limit Consultant’s right to discuss Consultant’s engagement with the Client or report possible violations of law or regulation with the Equal Employment Opportunity Commission, the United States Department of Labor, the National Labor Relations Board, the Securities and Exchange Commission, or other federal government agency or similar state or local
agency or to discuss the terms and conditions of Consultant’s engagement with others to the extent expressly permitted by applicable provisions of law or regulation, including but not limited to “whistleblower” statutes or other similar provisions that protect such disclosure. Further, notwithstanding the foregoing, pursuant to 18 U.S.C. Section 1833(b), Consultant shall not be held criminally or civilly liable under any Federal or State trade secret law for the disclosure of a trade secret that: (1) is made in confidence to a Federal, State, or local government official, either directly or indirectly, or to an attorney, and solely for the purpose of reporting or investigating a suspected violation of law; or (2) is made in a complaint or other document filed in a lawsuit or other proceeding, if such filing is made under seal. Consultant’s duty of confidentiality under this Agreement does not amend or abrogate in any manner Consultant’s continuing duties under any prior agreement between Consultant and the Client.
14.1Term. The term of this Agreement and the “Consulting Period” is from the Consulting Agreement Effective Date set forth above until the earlier of (i) the closing of a Change in Control or Corporate Transaction (each as defined in the Plan); and (ii) December 31, 2025 (which date may be extended on a monthly basis by the Company in its sole discretion). The Consulting Period may be earlier terminated as provided in this Agreement, or further extended by the parties by written agreement.
(a) Automatic Termination. If Consultant fails to timely return the Separation Agreement to the Company, then this Agreement will automatically terminate effective as of the date after the Consultant’s deadline to execute the Separation Agreement, and no benefits will be due to Consultant under this Agreement. If Consultant revokes his or her acceptance of the Separation Agreement as described therein and if applicable, then this Agreement will automatically terminate on the day of such revocation and no benefits will be due to Consultant under this Agreement.
(b) Termination upon Material Breach. The Client may terminate this Agreement before its expiration immediately if, Consultant materially breaches the Agreement. The parties agree that a “Material Breach” by Consultant shall occur if he: (i) breaches any material obligations of this Agreement, the Separation Agreement or the Confidential Information Agreement, or (ii) violates local, state, or federal laws. For avoidance of doubt, it shall not constitute a material breach if Consultant becomes employed or otherwise provides services to or on behalf of an entity other than the Client during the Consulting Period so long as any such activity does not conflict with Consultant’s continuing obligations to Client under the Confidential Information Agreement or the Separation Agreement.
14.3Effect of Termination. Upon any termination or expiration of this Agreement, Consultant (i) shall immediately discontinue all use of Client’s Confidential Information delivered under this Agreement; (ii) shall delete any such Client Confidential Information from Consultant’s computer storage or any other media, including, but not limited to, online and off-line libraries; and (iii) shall return to Client, or, at Client’s option, destroy, all copies of such Confidential Information then in Consultant’s possession (other than the laptop as set forth in the Separation
Agreement). Upon the end of the Term, the vesting of any Equity Awards shall cease immediately and Consultant’s right to exercise will be as set forth in the Equity Documents and, if applicable, the Separation Agreement.
14.4Survival. The rights and obligations contained in Sections 3-6, 8-9, 12, 13, 14.3, 14.4, and 15-21 will survive any termination or expiration of this Agreement.
15.Successors and Assigns. Consultant may not subcontract or otherwise delegate his obligations under this Agreement without Client’s prior written consent. Client may assign this Agreement. Subject to the foregoing, this Agreement will be for the benefit of Client’s successors and assigns, and will be binding on Consultant’s subcontractors or delegates.
16.Notices. Any notice required or permitted by this Agreement shall be in writing and shall be delivered as follows with notice deemed given as indicated: (i) by overnight courier upon written verification of receipt; or (ii) by telecopy, email, or facsimile transmission upon acknowledgment of receipt of electronic transmission. Notice shall be sent to the addresses set forth below or such other address as either party may specify in writing.
17.Governing Law. This Agreement shall be governed in all respects by the laws of the state or commonwealth from which Consultant primarily provides services under this Agreement (“Consultant State”), as such laws are applied to agreements entered into and to be performed entirely within the Consultant State between residents of the Consultant State. Any suit involving this Agreement shall be brought in a court sitting in the Consultant State. The parties agree that venue shall be proper in such courts, and that such courts will have personal jurisdiction over them.
18.Severability. Should any provisions of this Agreement be held by a court of law to be illegal, invalid or unenforceable, the legality, validity and enforceability of the remaining provisions of this Agreement shall not be affected or impaired thereby.
19.Waiver. The waiver by Client of a breach of any provision of this Agreement by Consultant shall not operate or be construed as a waiver of any other or subsequent breach by Consultant.
20.Injunctive Relief for Breach. Consultant’s obligations under this Agreement are of a unique character that gives them particular value; breach of any of such obligations will result in irreparable and continuing damage to Client for which there will be no adequate remedy at law; and, in the event of such breach, Client will be entitled to injunctive relief and/or a decree for specific performance, and such other and further relief as may be proper (including monetary damages if appropriate and attorneys’ fees).
21.Entire Agreement. This Agreement is being entered into as part of the Separation Agreement between the Client and Consultant, and is contingent upon Consultant’s execution and non-revocation of the Separation Agreement. This Agreement, the Separation Agreement, and the exhibits to the Separation Agreement, constitute the entire understanding of the parties relating to the subject matter and supersede any previous oral or written communications, representations, understanding, or agreement between the parties concerning such subject matter. This Agreement shall not be changed, modified, supplemented or amended except by express written agreement signed by Consultant and the Client. The parties have entered into separate agreements related to
Consultant’s previous employment relationship with Client, including but not limited to the Separation Agreement. These separate agreements govern the previous employment relationship between Consultant and Client, have or may have provisions that survive termination of Consultant’s relationship with Client (including under this Agreement), may be amended or superseded without regard to this Agreement, and are enforceable according to their terms without regard to the enforcement provision of this Agreement.
[signatures to follow on next page]
In Witness Whereof, the parties have executed this Agreement effective as of the date first written above.
Clearside Biomedical, Inc.
By: /s/George Lasezkay
George Lasezkay
President and Chief Executive Officer
Agreed to and Accepted:
/s/Victor Chone
Victor Chong
July 19, 2025
Date
EX-31.1
Exhibit 31.1
CERTIFICATION OF PRINCIPAL EXECUTIVE OFFICER
PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, George Lasezkay, certify that:
1.I have reviewed this Quarterly Report on Form 10-Q for the period ended June 30, 2025 of Clearside Biomedical, Inc. (the “registrant”);
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting.
5.The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
Date: August 8, 2025
|
|
|
/s/ George Lasezkay, Pharm.D., J.D. |
|
George Lasezkay, Pharm. D., J.D. |
|
President and Chief Executive Officer |
|
(principal executive officer) |
EX-31.2
Exhibit 31.2
CERTIFICATION OF PRINCIPAL FINANCIAL OFFICER
PURSUANT TO SECTION 302 OF THE SARBANES-OXLEY ACT OF 2002
I, Charles A. Deignan, certify that:
1.I have reviewed this Quarterly Report on Form 10-Q for the period ended June 30, 2025 of Clearside Biomedical, Inc. (the “registrant”);
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b)Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c)Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
(d)Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting.
5.The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
(a)All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
(b)Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
Date: August 8, 2025
|
|
|
/s/ Charles A. Deignan |
|
Charles A. Deignan |
|
Chief Financial Officer |
|
(principal financial officer) |
EX-32.1
Exhibit 32.1
CERTIFICATIONS OF
PRINCIPAL EXECUTIVE OFFICER AND PRINCIPAL FINANCIAL OFFICER
PURSUANT TO 18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
Pursuant to the requirement set forth in Rule 13a-14(b) of the Securities Exchange Act of 1934, as amended, (the “Exchange Act”) and Section 1350 of Chapter 63 of Title 18 of the United States Code (18 U.S.C. §1350), George Lasezkay, President and Chief Executive Officer of Clearside Biomedical, Inc. (the “Company”), and Charles A. Deignan, Chief Financial Officer of the Company, each hereby certifies that, to the best of his knowledge:
1.The Company’s Quarterly Report on Form 10-Q for the period ended June 30, 2025, to which this Certification is attached as Exhibit 32.1 (the “Periodic Report”), fully complies with the requirements of Section 13(a) or Section 15(d) of the Exchange Act; and
2.The information contained in the Periodic Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
IN WITNESS WHEREOF, the undersigned have set their hands hereto as of the 8th day of August, 2025.
|
|
|
/s/ George Lasezkay, Pharm. D., J.D. |
|
/s/ Charles. A. Deignan |
George Lasezkay, Pharm. D., J.D. |
|
Charles A. Deignan |
President and Chief Executive Officer
(principal executive officer) |
|
Chief Financial Officer
(principal financial officer) |
* This certification accompanies the Form 10-Q to which it relates, is not deemed filed with the Securities and Exchange Commission and is not to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Exchange Act (whether made before or after the date of the Form 10-Q), irrespective of any general incorporation language contained in such filing.








